How hosts recognize bacteria
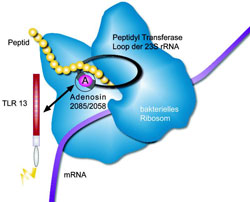
A bacterial 23S ribososmal (r) RNA segment activates the Toll-like receptor 13. This 23S rRNA segment is known to also bind antibiotics such as erythromycin (ery). Its central adenosine (A) 2085 in S. aureus and A2058 in E. coli 23S rRNA is mutated or methylated in ery resistant bacteria.<br>Design: Daniela Willemsen<br><br>
We are surrounded by bacteria, viruses, fungi and parasites. The fact that we nevertheless do not fall prey to infections is thanks to certain cellular sensor molecules such as toll-like receptors (TLR), which recognize the molecular structure of pathogens and intercede by ensuring an often completely unnoticeable elimination of the invaders.
Their immune-activating abilities were only detected in 1998, a discovery which was awarded with the Nobel Prize. Now, there are new findings published by an international Team of microbiologists in the research journal Science.
The international research team led by Prof. Dr. Carsten Kirschning of the Institute of Medical Microbiology at the University hospital Essen and the University of Duisburg-Essen and PD Dr. Hubertus Hochrein is examining the complex recognition of bacteria.
Our innate immune system is clever. Certain structures, which are characteristic of many microorganisms are recognized via TLR which trigger the necessary inflammatory response. However, the situation can become dangerous: since this receptor system is so sensitive, the immune response to serious infections may be over-exaggerated and miss the mark. Blood poisoning and frequently septic shock is the result.
Thus, the experts decided to take a closer look at the host recognition of Staphylococcus and E.coli bacteria, often the main agents of blood poisoning. They established that in the cell, TLR13 recognises the segment of bacterial ribosomal ribonucleic acid to which specific antibiotics such as Erythromycin also bind should the segment not have been altered by mutation or other modifications. The concrete ribonucleic acid is referred as 23S rRNA. Animal and human ribosomes do not bind Erythromycin to their own 28S rRNA because its structure resembles that of the 23S rRNA of resistant bacteria.
The new findings are significant for the treatment of bacterial infections and gaining a greater understanding of antibiotic resistance. In addition, these findings may be of help in the therapy of immunological overreactions and lead to new vaccination strategies.
The study was conducted by an international research team led by Prof. Dr. Carsten Kirschning of the Institute of Medical Microbiology at the University hospital, University of Duisburg-Essen, PD Dr. Hubertus Hochrein of Bavarian Nordic in Martinsried, Prof. Dr. Stefan Bauer of the Institute of Immunology at the Philipps University of Marburg and Prof. Dr. Hermann Wagner of the Institute of Medical Microbiology, Immunology and Hygiene at the Technical University of Munich. Further collaborating scientists came from Munich, Switzerland and Japan.
Publication: „TLR13 Recognizes Bacterial 23S rRNA Devoid of Erythromycin Resistance-Forming Modification.“ (Science Express)
Further information:
http://www.sciencemag.org/
● Prof. Dr. Carsten Kirschning, Tel: +49 201/723-1824, Cell: +49 1609/254-7309, carsten.kirschning@uk-essen.de
● Press officer of the University hospital Essen: Christine Harrell, Tel. +49 201/723-3107, christine.harrell@uk-essen.de
Editing: Carmen Tomlik, Tel. +49 203/379-1489
Translation: Delia Cosgrove, Tel. +49 201/723-4257, delia.cosgrove@uni-due.de
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

High-energy-density aqueous battery based on halogen multi-electron transfer
Traditional non-aqueous lithium-ion batteries have a high energy density, but their safety is compromised due to the flammable organic electrolytes they utilize. Aqueous batteries use water as the solvent for…

First-ever combined heart pump and pig kidney transplant
…gives new hope to patient with terminal illness. Surgeons at NYU Langone Health performed the first-ever combined mechanical heart pump and gene-edited pig kidney transplant surgery in a 54-year-old woman…

Biophysics: Testing how well biomarkers work
LMU researchers have developed a method to determine how reliably target proteins can be labeled using super-resolution fluorescence microscopy. Modern microscopy techniques make it possible to examine the inner workings…





















