Just Add Water: How Scientists Are Using Silicon to Produce Hydrogen on Demand
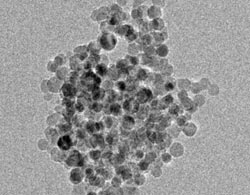
Transmission electron microscopy image showing spherical silicon nanoparticles about 10 nanometers in diameter. These particles, created in a UB lab, react with water to quickly produce hydrogen, according to new UB research. Credit: Swihart Research Group, University at Buffalo. <br>
Super-small particles of silicon react with water to produce hydrogen almost instantaneously, according to University at Buffalo researchers.
In a series of experiments, the scientists created spherical silicon particles about 10 nanometers in diameter. When combined with water, these particles reacted to form silicic acid (a nontoxic byproduct) and hydrogen — a potential source of energy for fuel cells.
The reaction didn’t require any light, heat or electricity, and also created hydrogen about 150 times faster than similar reactions using silicon particles 100 nanometers wide, and 1,000 times faster than bulk silicon, according to the study.
The findings appeared online in Nano Letters on Jan. 14. The scientists were able to verify that the hydrogen they made was relatively pure by testing it successfully in a small fuel cell that powered a fan.
“When it comes to splitting water to produce hydrogen, nanosized silicon may be better than more obvious choices that people have studied for a while, such as aluminum,” said researcher Mark T. Swihart, UB professor of chemical and biological engineering and director of the university’s Strategic Strength in Integrated Nanostructured Systems.
“With further development, this technology could form the basis of a ‘just add water’ approach to generating hydrogen on demand,” said researcher Paras Prasad, executive director of UB’s Institute for Lasers, Photonics and Biophotonics (ILPB) and a SUNY Distinguished Professor in UB’s Departments of Chemistry, Physics, Electrical Engineering and Medicine. “The most practical application would be for portable energy sources.”
Swihart and Prasad led the study, which was completed by UB scientists, some of whom have affiliations with Nanjing University in China or Korea University in South Korea. Folarin Erogbogbo, a research assistant professor in UB’s ILPB and a UB PhD graduate, was first author.
The speed at which the 10-nanometer particles reacted with water surprised the researchers. In under a minute, these particles yielded more hydrogen than the 100-nanometer particles yielded in about 45 minutes. The maximum reaction rate for the 10-nanometer particles was about 150 times as fast.
Swihart said the discrepancy is due to geometry. As they react, the larger particles form nonspherical structures whose surfaces react with water less readily and less uniformly than the surfaces of the smaller, spherical particles, he said.
Though it takes significant energy and resources to produce the super-small silicon balls, the particles could help power portable devices in situations where water is available and portability is more important than low cost. Military operations and camping trips are two examples of such scenarios.
“It was previously unknown that we could generate hydrogen this rapidly from silicon, one of Earth’s most abundant elements,” Erogbogbo said. “Safe storage of hydrogen has been a difficult problem even though hydrogen is an excellent candidate for alternative energy, and one of the practical applications of our work would be supplying hydrogen for fuel cell power. It could be military vehicles or other portable applications that are near water.”
“Perhaps instead of taking a gasoline or diesel generator and fuel tanks or large battery packs with me to the campsite (civilian or military) where water is available, I take a hydrogen fuel cell (much smaller and lighter than the generator) and some plastic cartridges of silicon nanopowder mixed with an activator,” Swihart said, envisioning future applications. “Then I can power my satellite radio and telephone, GPS, laptop, lighting, etc. If I time things right, I might even be able to use excess heat generated from the reaction to warm up some water and make tea.”
Media Contact Information
Charlotte Hsu
Media Relations Manager, Architecture, Economic Development, Sciences, Urban and Regional Planning
Tel: 716-645-4655
chsu22@buffalo.edu
Media Contact
More Information:
http://www.buffalo.eduAll latest news from the category: Power and Electrical Engineering
This topic covers issues related to energy generation, conversion, transportation and consumption and how the industry is addressing the challenge of energy efficiency in general.
innovations-report provides in-depth and informative reports and articles on subjects ranging from wind energy, fuel cell technology, solar energy, geothermal energy, petroleum, gas, nuclear engineering, alternative energy and energy efficiency to fusion, hydrogen and superconductor technologies.
Newest articles

High-energy-density aqueous battery based on halogen multi-electron transfer
Traditional non-aqueous lithium-ion batteries have a high energy density, but their safety is compromised due to the flammable organic electrolytes they utilize. Aqueous batteries use water as the solvent for…

First-ever combined heart pump and pig kidney transplant
…gives new hope to patient with terminal illness. Surgeons at NYU Langone Health performed the first-ever combined mechanical heart pump and gene-edited pig kidney transplant surgery in a 54-year-old woman…

Biophysics: Testing how well biomarkers work
LMU researchers have developed a method to determine how reliably target proteins can be labeled using super-resolution fluorescence microscopy. Modern microscopy techniques make it possible to examine the inner workings…





















