Parasite manipulates algal metabolism for its own benefit
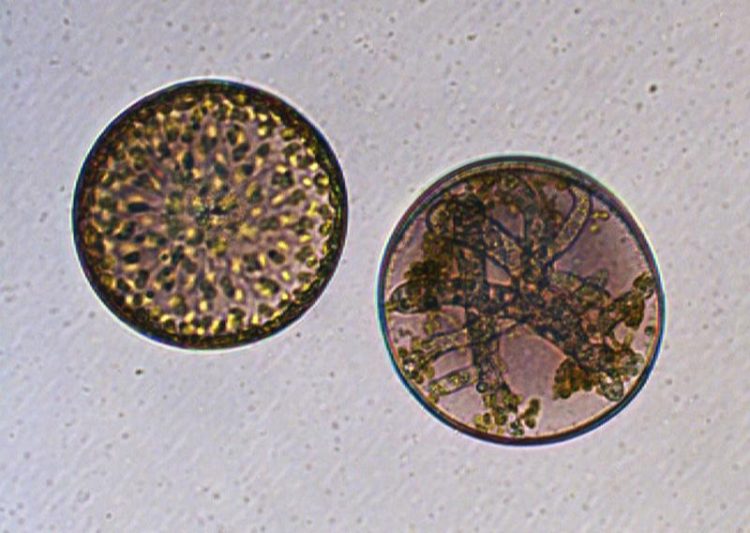
Healthy (left) and infected (right) diatom cell: In the cell on the right, the parasitic oomycete has sucked all nutrients and modulated the algal metabolome to generate its own reproductive form. Marine Vallet / Max Planck Institute for Chemical Ecology
Microalgae can form massive assemblages in oceans, attracting many opportunistic organisms; these are capable of eliminating the entire algal population within a short time.
However, the underlying mechanisms of this watery arms race are largely unknown. In a new publication in Nature Communications, researchers from the Max Planck Institute for Chemical Ecology and the universities of Jena and Frankfurt show that a pathogenic fungus alters the metabolism of its host unicellular algae, for its own purposes: the small bioactive substances that are formed in the process benefit the fungi’s own propagation while preventing the algae from proliferating. Eventually the algae shrink and die. (Nature Communications, October 2019, DOI 10.1038/s41467-019-12908-w).
Egg fungi – oomycetes – are known to cause many dangerous diseases in plants and animals. They also infest fish and algae, but the relationship between these microorganisms and algae is still poorly understood. Up to now, why some species first massively reproduce in the oceans and then disappear again has been unclear.
One hypothesis is that microorganisms produce chemical signaling substances which are regulating the interactions, such as defense, mating and communication, between living organisms. In order to identify such substances, the team of scientists established a laboratory system in which the egg fungus Lagenisma coscinodisci infects a marine diatom under controlled conditions.
Researchers found that during the infection process, two new substances were formed: both were so-called carbolines from the class of alkaloids which also includes nicotine and caffeine. Large amounts of these compounds accumulate after oomycete infection.
“The identity of the substances surprised us, as it has been described in diatoms for the first time and their induction during infection was conserved in the whole population of cells,” explains Marine Vallet, one of the two main authors of the new study. Interestingly, although these two substances benefited the oomycete, they were detrimental to the algae, ultimately killing it.
It is not easy to investigate such a system, because these fungal pests kill their host within just a few hours. “Oomycetes are known to take various forms: They are often found only as tiny spores in their host. Sometimes they do not cause any harm at all and are ‘asleep’ in their host, at other occasions they can cause mass killing of the cells. These processes result in highly dynamic fluctuation of dominant species in the oceans,” says Tim Baumeister, another main author, describing the initial challenges that the researchers faced.
Using high-resolution mass spectrometric methods to separate and identify small substances from complex mixtures, combined with microscopic techniques, scientists were able to identify the active compounds produced by a single algal cell.
“One has to bear in mind that a single cell is 30 times smaller than a pinhead and that the concentrations of all substances are very low – to show the chemistry in one single cell is a major technical achievement,” explains Georg Pohnert, head of the Max Planck Fellow Group Interactions in Plankton Communities and Chair of Instrumental Analytics at Friedrich Schiller University Jena.
Not all diatoms are equally vulnerable to attack, so how do certain diatoms defend themselves? These and many other unresolved questions remain to be addressed in follow-up studies The team is especially interested in identifying signaling substances involved in diatom interactions with their environment. “Our ocean is a treasure to protect, there are still so many fantastic discoveries awaiting us there,” concludes Marine Vallet. [KG/AO]
Contact and Picture Requests:
Angela Overmeyer M.A., Max Planck Institute for Chemical Ecology, Hans-Knöll-Str. 8, 07743 Jena, Germany, Tel. +49 3641 57-2110, E-Mail overmeyer@ice.mpg.de
Download of the video and high-resolution images via http://www.ice.mpg.de/ext/downloads2019.html
Prof. Dr. Georg Pohnert, Institute for Inorganic and Analytical Chemistry, Friedrich Schiller University Jena, Lessingstrasse 8, 07743 Jena, Germany, Tel. +49 3641 948170, E-Mail Georg.Pohnert@uni-jena.de
Dr. Marine Vallet, Max Planck Institute for Chemical Ecology, Hans-Knöll-Str. 8, 07745 Jena, Tel. +49 3641 57-2501 E-Mail: mvallet@ice.mpg.de
Vallet, M., Baumeister, T. U. H., Kaftan, F., Grabe, V., Buaya, A., Thines, M., Svatoš, A., Pohnert, G. (2019). The oomycete Lagenisma coscinodisci hijacks host alkaloid synthesis during infection of a marine diatom. Nature Communications. DOI 10.1038/s41467-019-12908-w
https://doi.org/10.1038/s41467-019-12908-w
https://www.ice.mpg.de/ext/index.php?id=plankton-interaction&L=0 – Max Planck Fellow Group Plankton Community Interaction
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

A universal framework for spatial biology
SpatialData is a freely accessible tool to unify and integrate data from different omics technologies accounting for spatial information, which can provide holistic insights into health and disease. Biological processes…

How complex biological processes arise
A $20 million grant from the U.S. National Science Foundation (NSF) will support the establishment and operation of the National Synthesis Center for Emergence in the Molecular and Cellular Sciences (NCEMS) at…

Airborne single-photon lidar system achieves high-resolution 3D imaging
Compact, low-power system opens doors for photon-efficient drone and satellite-based environmental monitoring and mapping. Researchers have developed a compact and lightweight single-photon airborne lidar system that can acquire high-resolution 3D…





















