Early organic carbon got deep burial in mantle
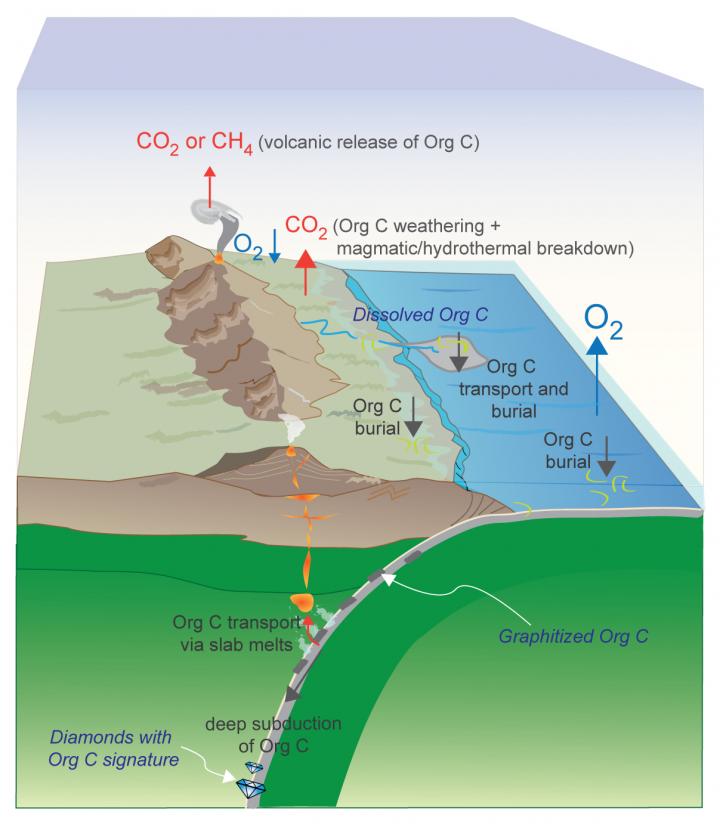
This schematic depicts the efficient deep subduction of organic (reduced) carbon, a process that could have locked significant amounts of carbon in Earth's mantle and resulted in a higher percentage of atmospheric oxygen. Based on new high-pressure, high-temperature experiments, Rice University petrologists argue that the long-term sequestration of organic carbon from this process began as early as 2.5 billion years ago and helped bring about a well-known buildup of oxygen in Earth's atmosphere -- the "Great Oxidation Event" -- about 2.4 billion years ago. Image courtesy of R. Dasgupta/Rice University
Rice University petrologists who recreated hot, high-pressure conditions from 60 miles below Earth's surface have found a new clue about a crucial event in the planet's deep past.
Their study describes how fossilized carbon — the remains of Earth's earliest single-celled creatures — could have been subsumed and locked deep in Earth's interior starting around 2.4 billion years ago — a time when atmospheric oxygen rose dramatically. The paper appears online this week in the journal Nature Geoscience.
“It's an interesting concept, but in order for complex life to evolve, the earliest form of life needed to be deeply buried in the planet's mantle,” said Rajdeep Dasgupta, a professor of Earth science at Rice. “The mechanism for that burial comes in two parts. First, you need some form of plate tectonics, a mechanism to carry the carbon remains of early life-forms back into Earth. Second, you need the correct geochemistry so that organic carbon can be carried deeply into Earth's interior and thereby removed from the surface environment for a long time.”
At issue is what caused the “great oxidation event,” a steep increase in atmospheric oxygen that is well-documented in countless ancient rocks. The event is so well-known to geologists that they often simply refer to it as the “GOE.” But despite this familiarity, there's no scientific consensus about what caused the GOE. For example, scientists know Earth's earliest known life, single-celled cyanobacteria, drew down carbon dioxide from the atmosphere and released oxygen. But the appearance of early life has been pushed further and further into the past with recent fossil discoveries, and scientists now know that cyanobacteria were prevalent at least 500 million years before the GOE.
“Cyanobacteria may have played a role, but the GOE was so dramatic — oxygen concentration increased as much as 10,000 times — that cyanobacteria by themselves could not account for it,” said lead co-author Megan Duncan, who conducted the research for her Ph.D. dissertation at Rice. “There also has to be a mechanism to remove a significant amount of reduced carbon from the biosphere, and thereby shift the relative concentration of oxygen within the system,” she said.
Removing carbon without removing oxygen requires special circumstances because the two elements are prone to bind with one another. They form one of the key components of the atmosphere — carbon dioxide — as well as all types of carbonate rocks.
Dasgupta and Duncan found that the chemical composition of the “silicate melt” — subducting crustal rock that melts and rises back to the surface through volcanic eruptions — plays a crucial role in determining whether fossilized organic carbon, or graphite, sinks into the mantle or rises back to the surface through volcanism.
Duncan, now a research scientist at the Carnegie Institution in Washington, D.C., said the study is the first to examine the graphite-carrying capacity of a type of melt known as rhyolite, which is commonly produced deep in the mantle and carries significant amounts of carbon to the volcanoes. She said the graphite-carrying capacity of rhyolitic rock is crucial because if graphite is prone to hitching a ride back to the surface via extraction of rhyolitic melt, it would not have been buried in sufficient quantities to account for the GOE.
“Silicate composition plays an important role,” she said. “Scientists have previously looked at carbon-carrying capacities in compositions that were much more magnesium-rich and silicon-poor. But the compositions of these rhyolitic melts are high in silicon and aluminum and have very little calcium, magnesium and iron. That matters because calcium and magnesium are cations, and they change the amount of carbon you can dissolve.”
Dasgupta and Duncan found that rhyolitic melts could dissolve very little graphite, even when very hot.
“That was one of our motivations,” said Dasgupta, professor of Earth science. “If subduction zones in the past were very hot and produced a substantial amount of melt, could they completely destabilize organic carbon and release it back to the surface?
“What we showed was that even at very, very high temperatures, not much of this graphitic carbon dissolves in the melt,” he said. “So even though the temperature is high and you produce a lot of melt, this organic carbon is not very soluble in that melt, and the carbon gets buried in the mantle as a result.
“What is neat is that with the onset and the expected tempo of crustal burial into the deep mantle starting just prior to the GOE, and with our experimental data on the efficiency of deep burial of reduced carbon, we could model the expected rise of atmospheric oxygen across the GOE,” Dasgupta said.
The research supports the findings of a 2016 paper by fellow Rice petrologist Cin-Ty Lee and colleagues that suggested that plate tectonics, continent formation and the appearance of early life were key factors in the development of an oxygen-rich atmosphere on Earth.
Duncan, who increasingly focuses on exoplanetary systems, said the research could provide important clues about what scientists should look for when evaluating which exoplanets could support life.
###
The research is supported by the National Science Foundation and the Deep Carbon Observatory.
High-resolution IMAGES are available for download at:
http://eol.
CAPTION: Earth's atmosphere, as seen in 2003 from the International Space Station, hasn't always contained large amounts of oxygen. Petrologists from Rice University and the Carnegie Institution recreated hot, high-pressure conditions from 60 miles below Earth's surface in search of new clues about the “great oxidation event” that added large amounts of oxygen to the atmosphere around 2.4 billion years ago. (Photo courtesy of ISS Expedition 7 Crew, EOL, NASA)
http://news.
CAPTION: Megan Duncan (Photo by Jeff Fitlow/Rice University)
http://news.
CAPTION: Rajdeep Dasgupta (Photo by Jeff Fitlow/Rice University)
http://news.
CAPTION: This schematic depicts the efficient deep subduction of organic (reduced) carbon, a process that could have locked significant amounts of carbon in Earth's mantle and resulted in a higher percentage of atmospheric oxygen. Based on new high-pressure, high-temperature experiments, Rice University petrologists argue that the long-term sequestration of organic carbon from this process began as early as 2.5 billion years ago and helped bring about a well-known buildup of oxygen in Earth's atmosphere — the “Great Oxidation Event” — about 2.4 billion years ago. (Image courtesy of R. Dasgupta/Rice University)
The DOI of the Nature Geoscience paper is: 10.1038/ngeo2939
A copy of the paper is available at: http://dx.
More information is available at:
Experimental Petrology Rice Team
http://dasgupta.
Similar research from Rice:
Study: Earth's carbon points to planetary smashup — Sept. 5, 2016
http://news.
Oxygen atmosphere recipe = tectonics + continents + life — May 16, 2016
http://news.
Going deep to study long-term climate evolution — Oct. 31, 2013
http://news.
Earth scientist Dasgupta lands NSF CAREER Award — March 21, 2013
http://news.
Magma in mantle has deep impact — Jan. 9, 2013
http://news.
Located on a 300-acre forested campus in Houston, Rice University is consistently ranked among the nation's top 20 universities by U.S. News & World Report. Rice has highly respected schools of Architecture, Business, Continuing Studies, Engineering, Humanities, Music, Natural Sciences and Social Sciences and is home to the Baker Institute for Public Policy. With 3,879 undergraduates and 2,861 graduate students, Rice's undergraduate student-to-faculty ratio is 6-to-1. Its residential college system builds close-knit communities and lifelong friendships, just one reason why Rice is ranked No. 1 for happiest students and for lots of race/class interaction by the Princeton Review. Rice is also rated as a best value among private universities by Kiplinger's Personal Finance. To read “What they're saying about Rice,” go to http://tinyurl.
Media Contact
All latest news from the category: Earth Sciences
Earth Sciences (also referred to as Geosciences), which deals with basic issues surrounding our planet, plays a vital role in the area of energy and raw materials supply.
Earth Sciences comprises subjects such as geology, geography, geological informatics, paleontology, mineralogy, petrography, crystallography, geophysics, geodesy, glaciology, cartography, photogrammetry, meteorology and seismology, early-warning systems, earthquake research and polar research.
Newest articles

High-energy-density aqueous battery based on halogen multi-electron transfer
Traditional non-aqueous lithium-ion batteries have a high energy density, but their safety is compromised due to the flammable organic electrolytes they utilize. Aqueous batteries use water as the solvent for…

First-ever combined heart pump and pig kidney transplant
…gives new hope to patient with terminal illness. Surgeons at NYU Langone Health performed the first-ever combined mechanical heart pump and gene-edited pig kidney transplant surgery in a 54-year-old woman…

Biophysics: Testing how well biomarkers work
LMU researchers have developed a method to determine how reliably target proteins can be labeled using super-resolution fluorescence microscopy. Modern microscopy techniques make it possible to examine the inner workings…





















