p53 cuts off invading cancer cells
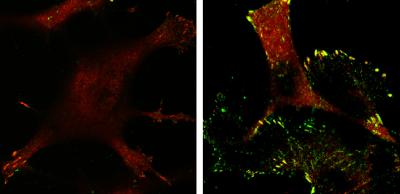
Tumor cells expressing p53 (left) have fewer focal adhesions (red) and lower levels of active p130Cas (green)—rendering them noninvasive—compared with p53-deficient tumor cells (right). Credit: Yamauchi et al., 2014
Sometimes oncogenes manage to initiate tumor development in the presence of p53, but, even then, the tumor suppressor doesn't give up and focuses its efforts instead on limiting the tumor's ability to invade and metastasize. A study in The Journal of Cell Biology uncovers one way that p53 acts to prevent cancer cell invasion.
A team of researchers, led by Keiko Kawauchi from the Mechanobiology Institute at the National University of Singapore, studied cells that had been transformed into cancer cells by Ras, the most common oncogene in human cancer.
They compared Ras-transformed cells with and without p53 and observed that those expressing p53 were less invasive and formed fewer focal adhesions, the molecular linkages that connect the structural scaffolding within the cell to the extracellular matrix that surrounds the cell.
The researchers found that p53 limits invasion by initiating a chain of events that ultimately prevents the formation of lamellipodia, cell membrane protrusions that spur cell movement and invasion. p53 activates a mitochondrial protease called Omi, which is then released into the cytosol of the cell when Ras causes mitochondria to fragment.
Omi cleaves actin filaments in the cytoskeleton, and the decrease in actin suppresses the activity of p130Cas, a focal adhesion signaling protein that promotes the formation of lamellipodia. With low levels of active p130Cas, cells don't form lamellipodia and are therefore less able to invade.
“Actin remodeling is a signal that prevents cell invasion,” explains Kawauchi. “Most research has focused on how p53 prevents metastasis by regulating epithelial-to-mesenchymal transitions,” a biological process by which cells gain migratory and invasive properties. In contrast, says Kawauchi, the new findings help explain how p53 affects the cytoskeletal processes within the cell that drive invasion.
Yamauchi, S., et al. 2014. J. Cell Biol. doi:10.1083/jcb.201309107
About The Journal of Cell Biology
The Journal of Cell Biology (JCB) is published by The Rockefeller University Press. All editorial decisions on manuscripts submitted are made by active scientists in conjunction with our in-house scientific editors. JCB content is posted to PubMed Central, where it is available to the public for free six months after publication. Authors retain copyright of their published works, and third parties may reuse the content for non-commercial purposes under a creative commons license. For more information, please visit http://www.jcb.org.
Research reported in the press release was supported by the National Research Foundation, Singapore; the Ministry of Education, Singapore; and the Kurata Memorial Hitachi Science and Technology Foundation, Japan.
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

A universal framework for spatial biology
SpatialData is a freely accessible tool to unify and integrate data from different omics technologies accounting for spatial information, which can provide holistic insights into health and disease. Biological processes…

How complex biological processes arise
A $20 million grant from the U.S. National Science Foundation (NSF) will support the establishment and operation of the National Synthesis Center for Emergence in the Molecular and Cellular Sciences (NCEMS) at…

Airborne single-photon lidar system achieves high-resolution 3D imaging
Compact, low-power system opens doors for photon-efficient drone and satellite-based environmental monitoring and mapping. Researchers have developed a compact and lightweight single-photon airborne lidar system that can acquire high-resolution 3D…





















