Electrocatalysis under the atomic force microscope
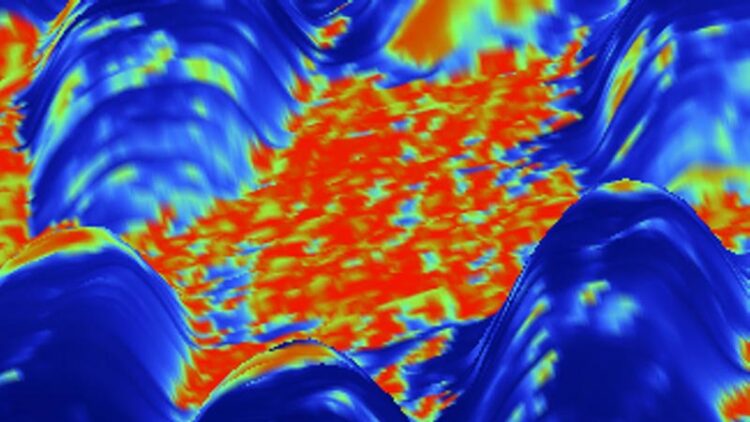
Electrocatalysis under the atomic force microscope The newly developed method was used to scan the surface of a bimetallic catalyst material in an aqueous medium. The figure shows an overlay of the current signal on a three-dimensional representation of the height image. This clearly shows island-like regions. The newly developed method was used to scan the surface of a bimetallic catalyst material in an aqueous medium. The figure shows an overlay of the current signal on a three-dimensional representation of the height image. This clearly shows island-like regions.
Credit: M.Munz/FHI/HZB
The simultaneous recording of height profiles, currents and frictional forces at solid-liquid interfaces allows exciting insights into electrocatalytically active materials.
To manage the energy transition, it will also be important to rapidly develop cheap and efficient materials that can be used to split water or CO2 by electrocatalysis. In this process, part of the electrical energy is stored in the chemical reaction products. The efficiency of such electrocatalysts depends largely on the nature of the electrode-electrolyte interfaces, i.e. the interfaces between the solid electrodes and the typically aqueous electrolyte. However, spatially resolved physical studies of such solid-liquid interfaces are still relatively scarce.
More insights with AFM
Dr Christopher S. Kley and his team have now developed a new approach to correlative atomic force microscopy (AFM). An extremely sharp tip is scanned across the surface and its height profile is recorded. By attaching the tip to the end of a miniaturised cantilever, the force interactions between the tip and the sample surface, including frictional forces, can be measured with high sensitivity. In addition, the electrical current flowing through the mechanical contact can be measured, provided a voltage is applied. “This allowed us to simultaneously determine the electrical conductivity, the mechanical-chemical friction and the morphological properties in situ (i.e. under the relevant liquid-phase conditions rather than in vacuum or in air),” emphasises Kley.
Copper-gold electrocatalyst
Using this method, the scientists now studied a nanostructured and bimetallic copper-gold electrocatalyst, in collaboration with Prof. Beatriz Roldán Cuenya from the Fritz-Haber-Institute (FHI). Among others, such materials are used in the electrocatalytic conversion of CO2 into energy carriers. “We were able to clearly identify islands of copper oxide with higher electrical resistance, but also grain boundaries and low-conductivity regions in the hydration layer where the catalyst surface comes into contact with the aqueous electrolyte,” says Dr Martin Munz, first author of the study.
Such results on catalyst-electrolyte interfaces help to optimise them in a targeted manner. “We can now observe how local electrochemical environments influence charge transfer at the interface,” says Kley.
Focus on solid-liquid interfaces
“However, our results are also of general interest to energy research, especially for the study of electrochemical conversion processes, which also play a role in battery systems.” Insights into solid-liquid interfaces can also be useful in completely different areas of research, such as understanding corrosion processes, nanosensor systems, and possibly addressing scientific queries in fluidics and environmental sciences, such as dissolution or deposition processes on metal surfaces exposed to water.
Note: This work was carried out within the framework of the CatLab project, where researchers from the HZB and the FHI of the MPG are working together, to develop thin-film catalysts for the energy transition.
Journal: Journal of the American Chemical Society
DOI: 10.1021/jacs.2c12617
Method of Research: Experimental study
Subject of Research: Not applicable
Article Title: Nanoscale Electron Transfer Variations at Electrocatalyst–Electrolyte Interfaces Resolved by in Situ Conductive Atomic Force Microscopy
Article Publication Date: 22-Feb-2023
COI Statement: none
Media Contact
Antonia Roetger
Helmholtz-Zentrum Berlin für Materialien und Energie
antonia.roetger@helmholtz-berlin.de
Office: 0049-308-062-43733
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles
Faster, more energy-efficient way to manufacture an industrially important chemical
Zirconium combined with silicon nitride enhances the conversion of propane — present in natural gas — needed to create in-demand plastic, polypropylene. Polypropylene is a common type of plastic found…

Energy planning in Ghana as a role model for the world
Improving the resilience of energy systems in the Global South. What criteria should we use to better plan for resilient energy systems? How do socio-economic, technical and climate change related…

Artificial blood vessels could improve heart bypass outcomes
Artificial blood vessels could improve heart bypass outcomes. 3D-printed blood vessels, which closely mimic the properties of human veins, could transform the treatment of cardiovascular diseases. Strong, flexible, gel-like tubes…




















