Visualizing single molecules in whole cells with a new spin
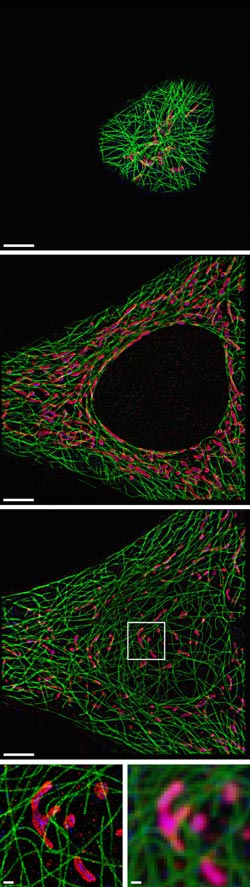
The researchers used their SDC-PAINT method to visualize the network of cytoskeletal microtubule filaments (green) and their proximity with two additional proteins called TOM20 (red) and HSP60 (blue). Each image shows the proteins in a different plane of the cell starting from the top, and the magnified images on the bottom compare the resolution achieved with SDC-PAINT (left) to that possible with conventional confocal microscopy (right). Credit: Florian Schueder, MPI/LMU
Cell biologists traditionally use fluorescent dyes to label and visualize cells and the molecules within them under a microscope. With different super-resolution microscopy methods, they can even light up single molecules and their complex interactions with one another. However, the microscopy hardware that allows them to do this is highly specialized and expensive and hence, relatively rare in laboratories around the world, and the operation of such microscopes is daunting, as it requires unique skills.
Ralf Jungmann, Ph.D., an alumnus of Harvard's Wyss Institute for Biologically Inspired Engineering and currently a Professor at the Ludwig Maximilian University (LMU) and the Max Planck Institute (MPI) of Biochemistry in Germany and Wyss Institute Core Faculty member Peng Yin, Ph.D., have been developing DNA-PAINT, a powerful molecular imaging technology that involves transient DNA-DNA interactions to accurately localize fluorescent dyes with super-resolution. However, although the researchers have demonstrated DNA-PAINT's potential by visualizing single biomolecules, such as proteins, in fixed cells at a fixed close distance, the technology could not yet be applied to investigate molecules deep inside of cells.
Now, Jungmann's and Yin's teams jointly report a solution to overcome this limitation. In their new study, they adapted DNA-PAINT technology to microscopes that are widespread among cell biology laboratories, called confocal microscopes, and that are used by researchers to image whole cells and thicker tissues at lower resolution. The MPI/Wyss Institute team demonstrates that the method can visualize a variety of different molecules, including combinations of different proteins, RNAs and DNA throughout the entire depth of whole cells at super-resolution. Published in Nature Communications, the approach could open the door for detailed single-molecule localization studies in many areas of cell research.
The DNA-PAINT approach attaches a DNA “anchor strand to the molecule of interest. Then a dye-labeled DNA “imager strand” with a complementary sequence transiently attaches to the anchor and produces a fluorescent signal, which occurs as a defined blinking event at single molecular sites. Because “blinking” is precisely tunable, molecules that are only nanometers apart from each other can be distinguished — at the higher resolution end of super-resolution.
“Our new approach, SDC-PAINT, integrates the versatile super-resolution capabilities of DNA-PAINT with the optical sectioning features of confocal microscopes. We thus created the means to explore the entire depth of a cell, and to visualize the molecules within it at the nanometer scale,” said Jungmann. The team mapped out the presence of different combinations of protein within whole cells and then went beyond that. “By diversifying our labeling approaches, we also visualized different types of individual biomolecules in the chromosome-containing nucleus, including sequences in the DNA, proteins bound to DNA or the membrane that encloses the nucleus, as well as nuclear RNAs,” adds Yin, who is also co-leader of the Wyss Institute's Molecular Robotics Initiative, and Professor of Systems Biology at Harvard Medical School. .
In principle, confocal microscopes use so-called pinholes to eliminate unwanted out-of-focus fluorescence from image planes above and below the focal plane. By scanning through the sample, plane after plane, researchers can gather the fluorescence signals emitted from molecule-bound dyes over the entire depth. Specifically, the MPI/Wyss Institute team developed the technique for “Spinning Disk Confocal” (SDC) microscopes that detect fluorescence signals from an entire plane all at once by sensing them through a rotating disc with multiple pinholes. Moreover, “to achieve 3D super-resolution, we placed an additional lens in the detection path, which allows us to archive sub-diffraction-limited resolution in the third dimension” said first author Florian Schueder, a Graduate Student working with Jungmann who also worked with Yin's Wyss Institute team as part of his master thesis.
“This addition can be easily customized by manufacturers of SDC microscopes; so we basically implement super-resolution microscopy without complex hardware changes to microscopes that are generally available to cell biologists from all venues of biomedical research. The approach thus has the potential to democratize super-resolution imaging throughout whole cells and tissues,” said Jungmann.
“With this important advance, super-resolution microscopy and DNA-PAINT could become more accessible to biomedical researchers, accelerating our insights into the function of individual molecules and the processes they control within cells,” said Wyss Institute Founding Director Donald Ingber, M.D., Ph.D., who is also the Judah Folkman Professor of Vascular Biology at HMS and the Vascular Biology Program at Boston Children's Hospital, as well as Professor of Bioengineering at SEAS.
###
Other authors on the study are past and present members of Yin's group, including Juanita Lara-Gutiérrez, Brian Beliveau, Ph.D., Sinem Saka, Ph.D., and Hiroshi Sasaki, Ph.D.; and Johannes Woehrstein, Maximilian Strauss and Heinrich Grabmayr, Ph.D., who are working with Jungmann. The study was funded by grants from the Wyss Institute for Biologically Inspired Engineering at Harvard University, the German Research Foundation's Emmy Noether Program, the European Research Council, LMU's Center for Nanoscience, the Max Planck Society and Max Planck Foundation, the National Institutes of Health and the Office of Naval Research.
MULTIMEDIA AVAILABLE
PRESS CONTACTS
Wyss Institute for Biologically Inspired Engineering at Harvard University
Benjamin Boettner, Benjamin.Boettner@wyss.harvard.edu, +1 617-432-8323
Max Planck Institute for Biochemistry
Christiane Menzfeld, menzfeld@biochem.mpg.de, +49 89-8578-2848
MULTIMEDIA CONTACT
Wyss Institute for Biologically Inspired Engineering at Harvard University
Seth Kroll, seth.kroll@wyss.harvard.edu, +1 617-432-7758
The Wyss Institute for Biologically Inspired Engineering at Harvard University uses Nature's design principles to develop bioinspired materials and devices that will transform medicine and create a more sustainable world. Wyss researchers are developing innovative new engineering solutions for healthcare, energy, architecture, robotics, and manufacturing that are translated into commercial products and therapies through collaborations with clinical investigators, corporate alliances, and formation of new startups. The Wyss Institute creates transformative technological breakthroughs by engaging in high risk research, and crosses disciplinary and institutional barriers, working as an alliance that includes Harvard's Schools of Medicine, Engineering, Arts & Sciences and Design, and in partnership with Beth Israel Deaconess Medical Center, Brigham and Women's Hospital, Boston Children's Hospital, Dana-Farber Cancer Institute, Massachusetts General Hospital, the University of Massachusetts Medical School, Spaulding Rehabilitation Hospital, Boston University, Tufts University, Charité – Universitätsmedizin Berlin, University of Zurich and Massachusetts Institute of Technology.
The Max Planck Institute of Biochemistry (MPIB) belongs to the Max Planck Society, an independent, non-profit research organization dedicated to top level basic research. As one of the largest Institutes of the Max Planck Society, 850 employees from 45 nations work here in the field of life sciences. In currently eight departments and about 25 research groups, the scientists contribute to the newest findings in the areas of biochemistry, cell biology, structural biology, biophysics and molecular science. The MPIB in Munich-Martinsried is part of the local life-science-campus where two Max Planck Institutes, a Helmholtz Center, the Gene-Center, several bio-medical faculties of two Munich universities and several biotech-companies are located in close proximity.
Harvard Medical School has more than 11,000 faculty working in 10 academic departments located at the School's Boston campus or in hospital-based clinical departments at 15 Harvard-affiliated teaching hospitals and research institutes: Beth Israel Deaconess Medical Center, Boston Children's Hospital, Brigham and Women's Hospital, Cambridge Health Alliance, Dana-Farber Cancer Institute, Harvard Pilgrim Health Care Institute, Hebrew SeniorLife, Joslin Diabetes Center, Judge Baker Children's Center, Massachusetts Eye and Ear/Schepens Eye Research Institute, Massachusetts General Hospital, McLean Hospital, Mount Auburn Hospital, Spaulding Rehabilitation Network and VA Boston Healthcare System.
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

A universal framework for spatial biology
SpatialData is a freely accessible tool to unify and integrate data from different omics technologies accounting for spatial information, which can provide holistic insights into health and disease. Biological processes…

How complex biological processes arise
A $20 million grant from the U.S. National Science Foundation (NSF) will support the establishment and operation of the National Synthesis Center for Emergence in the Molecular and Cellular Sciences (NCEMS) at…

Airborne single-photon lidar system achieves high-resolution 3D imaging
Compact, low-power system opens doors for photon-efficient drone and satellite-based environmental monitoring and mapping. Researchers have developed a compact and lightweight single-photon airborne lidar system that can acquire high-resolution 3D…





















