The unanticipated early origins of childhood brain cancer
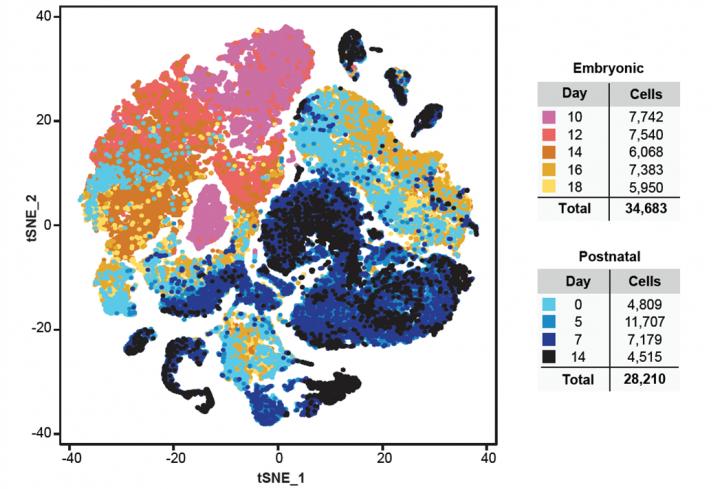
This is a graph-plot showing the relationship between 62,040 single cells isolated from the mouse cerebellum. Credit: Maria C. Vladoiu, Ibrahim El-Hamami and Laura K. Donovan.
Brain tumours are the leading cause of non-accidental death in children in Canada, but little is known about when these tumours form or how they develop. Researchers have recently identified the cells that are thought to give rise to certain brain tumours in children and discovered that these cells first appear in the embryonic stage of a mammal's development – far earlier than they had expected.
Their findings, published today in Nature, could lead the way to the discovery of better treatments to attack these lethal tumours.
“Progress in the development of more effective brain cancer treatments has been hampered in large part by the complex heterogeneity – or the variety of cells – within each tumour,” says Dr. Michael Taylor, Paediatric Neurosurgeon and Senior Scientist in Developmental and Stem Cell Biology at The Hospital for Sick Children (SickKids) and co-lead of the study.
“We recognized that new technologies could allow us to unravel some of this complexity, so we combined our expertise with McGill and OICR to approach this problem together.”
Using mouse models, the research group investigated the different types of normal brain cells and how they developed at various timepoints in the cerebellum of the brain – the most common location for childhood brain tumours to appear. They mapped the lineages of over 30 types of cells and identified normal cells that would later transform into cancerous cells, also known as the cells of origin.
To pinpoint these specific cells, the group relied on single cell sequencing technology, which allows researchers to look at individual cells more clearly than traditional sequencing methods.
In their investigation, the cells of origin were observed much earlier in fetal development than one would expect, says Taylor, who is also a Professor in the Departments of Surgery and Laboratory Medicine and Pathology at the University of Toronto and Co-lead of OICR's Brain Cancer Translational Research Institute.
“Our data show that in some cases, these tumours arise from cell populations and events that would occur in humans at six weeks in utero,” says Dr. Lincoln Stein, Head of Adaptive Oncology at OICR and co-lead of the study. “This means that the brain tumours may be starting long before they show in clinic, even before a woman may know she is pregnant.”
“The brain is extraordinarily complex. These findings are not only important for better understanding brain tumours but they will also allow us to learn more about these cells and how they work, in order to help children with neurodevelopmental delays. What we have accomplished as a team in this study brings hope for patients,” adds Dr. Nada Jabado, Paediatric Hemato-Oncologist and Senior Scientist in the Child Health and Human Development Program at the Research Institute of the McGill University Health Centre and co-lead of the study. Dr. Jabado is also a professor of Pediatrics and Human genetics at McGill University.
With this knowledge, researchers can now study the differences between the development of normal, healthy cells and the cells that will eventually give rise to cancerous cells.
###
This work was made possible through the 2017 Large-Scale Applied Research Project Competition Genomics and Precision Health supported by main funding agencies Génome Québec, the Canadian Institutes of Health Research (CIHR) and Genome Canada, as well as the Ontario Research Fund, SickKids Foundation, and the Montreal Children's Hospital Foundation. The work was also supported by the Ontario Institute for Cancer Research through funding provided by the Government of Ontario and the Stand Up To Cancer (SU2C) St. Baldrick's Pediatric Dream Team Translational Research Grant (SU2C-AACR-DT1113) and SU2C Canada Cancer Stem Cell Dream Team Research Funding (SU2C-AACR-DT-19-15) provided by the Government of Canada through Genome Canada and the Canadian Institutes of Health Research, with supplementary support from the Ontario Institute for Cancer Research through funding provided by the Government of Ontario. Stand Up To Cancer is a program of the Entertainment Industry Foundation administered by the American Association for Cancer Research.
About The Hospital for Sick Children (SickKids)
The Hospital for Sick Children (SickKids) is recognized as one of the world's foremost paediatric health-care institutions and is Canada's leading centre dedicated to advancing children's health through the integration of patient care, research and education. Founded in 1875 and affiliated with the University of Toronto, SickKids is one of Canada's most research-intensive hospitals and has generated discoveries that have helped children globally. Its mission is to provide the best in complex and specialized family-centred care; pioneer scientific and clinical advancements; share expertise; foster an academic environment that nurtures health-care professionals; and champion an accessible, comprehensive and sustainable child health system. SickKids is a founding member of Kids Health Alliance, a network of partners working to create a high quality, consistent and coordinated approach to pediatric health care that is centred around children, youth and their families. SickKids is proud of its vision for Healthier Children. A Better World.
About the Ontario Institute for Cancer Research (OICR)
OICR is a collaborative, not-for-profit research institute funded by the Government of Ontario. We conduct and enable high-impact translational cancer research to accelerate the development of discoveries for patients around the world while maximizing the economic benefit of this research for the people of Ontario. For more information visit http://www.
About the Research Institute of the MUHC
The Research Institute of the McGill University Health Centre (RI-MUHC) is a world-renowned biomedical and healthcare research centre. The Institute, which is affiliated with the Faculty of Medicine of McGill University, is the research arm of the McGill University Health Centre (MUHC) – an academic health centre located in Montreal, Canada, that has a mandate to focus on complex care within its community. The RI-MUHC supports over 420 researchers and close to 1,200 research trainees devoted to a broad spectrum of fundamental, clinical and health outcomes research at the Glen and the Montreal General Hospital sites of the MUHC. Its research facilities offer a dynamic multidisciplinary environment that fosters collaboration and leverages discovery aimed at improving the health of individual patients across their lifespan. The RI-MUHC is supported in part by the Fonds de recherche du Quebec – Sante; (FRQS). rimuhc.ca
Media contacts:
Jessamine Luck
The Hospital for Sick Children (SickKids)
416-813-7654 ext. 201436
jessamine.luck@sickkids.ca
Hal Costie
Ontario Institute for Cancer Research
647-260-7921
hal.costie@oicr.on.ca
Julie Robert
McGill University Health Centre
514-934-1934 ext. 71381
julie.robert@muhc.mcgill.ca
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

A universal framework for spatial biology
SpatialData is a freely accessible tool to unify and integrate data from different omics technologies accounting for spatial information, which can provide holistic insights into health and disease. Biological processes…

How complex biological processes arise
A $20 million grant from the U.S. National Science Foundation (NSF) will support the establishment and operation of the National Synthesis Center for Emergence in the Molecular and Cellular Sciences (NCEMS) at…

Airborne single-photon lidar system achieves high-resolution 3D imaging
Compact, low-power system opens doors for photon-efficient drone and satellite-based environmental monitoring and mapping. Researchers have developed a compact and lightweight single-photon airborne lidar system that can acquire high-resolution 3D…





















