The 'Signal Cell' relaying microbiota signals discovered
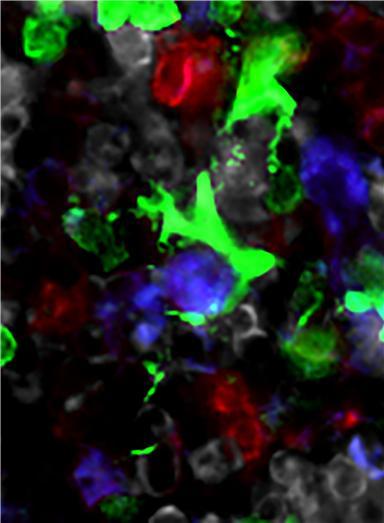
CX3CR1+ mononuclear cells (colored in green) are contacting hematopoietic progenitors (colored in purple) in the bone marrow. When CX3CR1+ mononuclear cells recognize the microbiota signals, they produce inflammatory cytokines which expedite the hematopoiesis. Credit: POHANG UNIVERSITY OF SCIENCE & TECHNOLOGY (POSTECH)
Professor Seung-Woo Lee, Research Professor Yunji Park, Master/PhD integrated program students, Seungwon Lee and Hyekang Kim of Division of Integrative Biosciences and Biotechnology from POSTECH described the mechanism how microbiota signals are sent to different organs.
Also, they utilized imaging research to prove that CX3CR+ mononuclear cells contact hematopoietic progenitors for the first time in history. Their research is introduced as a featured content in the journal of the American Society of Hematology, Blood.
Recent researches on microorganism concluded that microbiota control biological phenomenon not only in digestive tracts but also in lung, liver, brain, bone marrow and other organs. But, none of them were able to define a mechanism for relaying microbiota signals to entire body or for producing immune cells by receiving microbiota signals.
Professor Lee and his research team focused on the fact that the microbiota regulate the immune system of our body by controlling hematopoiesis in the bone marrow to produce white blood cells. In this process, the team discovered that the microbiota signal including bacterial DNA is transferred to the bone marrow through bloodstream and CX3CR1+ mononuclear cells in the bone marrow recognize this signal.
They explained that when CX3CR1+ mononuclear cells recognize microbiota signals, they release signal substances called cytokines which control and stimulate body's defense system through the signal transduction. They also explained that cytokines control the number of hematopoietic progenitors or stimulate differentiation into myeloid lineages to make blood cells.
Furthermore, they verified that CX3CR1+ mononuclear cells contact hematopoietic progenitors at the perivascular region and they play as a signal receiving microbiota signals.
They discovered the hematopoiesis control mechanism which is controlled by cytokines produced when CX3CR1+ mononuclear cells recognize microbiota signals transferred to the bone marrow.
Professor Seung-Woo Lee commented, “For the first time, our research describes the mechanism that had not been explained how microbiota regulate not only digestive tracts but also entire body response. It might be possible to apply this study to control immune response in other parts of a body or to treat cancer and inflammatory disease via microbiota signal pathway.
This study was financially supported by National Research Foundation of Korea, Regional Leading Research Center, and Korea Ministry of Science and ICT under BK21 Plus project.
Media Contact
Jinyoung Huh
jyhuh@postech.ac.kr
82-542-792-415
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

A universal framework for spatial biology
SpatialData is a freely accessible tool to unify and integrate data from different omics technologies accounting for spatial information, which can provide holistic insights into health and disease. Biological processes…

How complex biological processes arise
A $20 million grant from the U.S. National Science Foundation (NSF) will support the establishment and operation of the National Synthesis Center for Emergence in the Molecular and Cellular Sciences (NCEMS) at…

Airborne single-photon lidar system achieves high-resolution 3D imaging
Compact, low-power system opens doors for photon-efficient drone and satellite-based environmental monitoring and mapping. Researchers have developed a compact and lightweight single-photon airborne lidar system that can acquire high-resolution 3D…





















