The molecular basis of learning
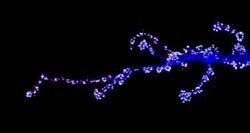
Motorneurons of fly larvae<br>Photo: AG Robert Kittel <br>
Somewhere between 100 and 1000 billion nerve cells can be found in the human brain. Every single one of them is in contact with 1000 other cells on average. The cells communicate with each other via synaptic connections and, in so doing, control our thoughts, actions, and feelings.
At the same time, this highly complex network manages to adapt to changed conditions as part of an ongoing process of self-organization. By doing this, it enables its host to cope with completely new and unexpected situations, and can even continue to perform when it has sustained damage.
Synaptic plasticity
Plasticity is the scientific term for this adaptability; synaptic plasticity is one of its sub-forms. Learning is based on this ability of the brain to forge new links, to strengthen existing ones, and to dismantle unused ones again. Scientists from the Institute of Physiology at the University of Würzburg have conducted research into which molecular changes take place at the synapses. The scientific journal Cell Reports covers their work in its latest issue.
“Synaptic plasticity has been known about for a long time. The Canadian psychologist Donald O. Hebb formulated a particularly influential hypothesis back in 1949 in his famous learning rule,” says Dr. Robert Kittel. Kittel is in charge of an Emmy Noether research group at the Institute of Physiology; he wrote the article for the journal’s publisher Cell Press with his colleagues Dmitrij Ljaschenko and Nadine Ehmann.
To put it briefly, according to Hebb, the more often a particular neuron is active at the same time as another neuron, the more preferentially the two neurons will react to each other – in line with the motto: what fires together, wires together.
Sights set on motorneurons
“Although we now know that synaptic activity and synapse development are intimately interconnected, our knowledge of the molecular mechanism of this relationship is far from complete,” says Robert Kittel. However, he and his team have now managed to uncover new information – with fly larvae, blue light, and a close look at the molecular details.
In their experiments, the scientists focused on the so-called “motorneurons” of fly larvae. These are nerve cells that contact the muscles and, via their synapses, give them the command to contract, for example. The dynamics at these points are major: “The muscle of the fly larva grows within a matter of days to a hundred times the size. This means that the synapses have to mirror that growth,” says Kittel.
There is another reason why fly motorneurons make an interesting research subject: in these motorneurons, glutamate is the transmitter that communicates information from the presynapse to the postsynapse – i.e. from the nerve cell to the muscle. In humans, glutamate is the main transmitter in the brain. It has been proven, therefore, that findings in relation to processes affecting larval motorneurons can readily be applied to humans – and other mammals.
Flashes of light on fly larvae
Using a sophisticated technique now known as “optogenetics”, the scientists succeeded in controlling the activity of the motorneurons. To achieve this, they inserted so-called channelrhodopsins into the cell walls. Channelrhodopsins are ion channels that are controlled by light. Cells use ion channels to conduct electrically charged particles through their cell membrane into the cell interior or out into the extracellular space. Nerve cells use this mechanism, for example, to transmit signals from sensations to the brain and to control muscles. One of the discoverers of this technique, Professor Georg Nagel, is a professor at the University of Würzburg’s Department of Molecular Plant Physiology and Biophysics. He collaborates with Kittel’s group on various projects.
“Given that fly larvae are practically transparent, we only had to expose them to blue light pulses to generate such activity in the neurons,” explains Kittel. The scientists spent 100 minutes stimulating the nerve cells in this manner to send signals to the muscles. They then examined which functional and molecular changes had taken place in the synapses.
The main findings
Following the intense activity of the motorneurons, the postsynaptic sensitivity, i.e. the sensitivity on the part of the muscles, was significantly elevated. There, the cell had integrated increased numbers of glutamate receptors of a specific subunit – so-called type 2A receptors. “However, this effect only occurred when nerve and muscle had been stimulated simultaneously,” says Kittel. This was a finding that grabbed the team’s attention – after all, it corresponds exactly to the Hebbian rule that states that connections are strengthened when the parties involved are active at the same time.
Conversely, these 2A subunits were removed again very quickly from synapses whose activity was not capable of evoking substantial activity on the part of the muscle. This, too, is a logical process: “If the quantity of receptors could only ever increase, the system would soon collapse,” says Kittel. So, there also has to be a way of “applying the brakes to the system.” If, therefore, a single synapse is the only one to send the muscle a command to contract, that is not normally enough to provoke a reaction. As a consequence, it is “punished” for acting on its own by the withdrawal of receptors.
These insights represent a “promising physiological concept,” says Kittel. They provide a picture of the molecular processes of synaptic plasticity and demonstrate how, in Hebbian learning, synapses mature and a sparse transmitter release controls the stabilization of the molecular composition of individual synapses. Armed with this knowledge of the basic mechanisms, he continues, it is now possible to research other places to see whether they apply equally there.
Hebbian Plasticity Guides Maturation of Glutamate Receptor Fields In Vivo. Dmitrij Ljaschenko, Nadine Ehmann, and Robert J. Kittel. Cell Reports, published online May 2. http://dx.doi.org/10.1016/j.celrep.2013.04.003
Contact
Dr. Robert Kittel, T: +49 (0)931 31-86046, robert.kittel@uni-wuerzburg.de
Media Contact
More Information:
http://www.uni-wuerzburg.deAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

A universal framework for spatial biology
SpatialData is a freely accessible tool to unify and integrate data from different omics technologies accounting for spatial information, which can provide holistic insights into health and disease. Biological processes…

How complex biological processes arise
A $20 million grant from the U.S. National Science Foundation (NSF) will support the establishment and operation of the National Synthesis Center for Emergence in the Molecular and Cellular Sciences (NCEMS) at…

Airborne single-photon lidar system achieves high-resolution 3D imaging
Compact, low-power system opens doors for photon-efficient drone and satellite-based environmental monitoring and mapping. Researchers have developed a compact and lightweight single-photon airborne lidar system that can acquire high-resolution 3D…





















