Helicobacter creates immune system blind spot
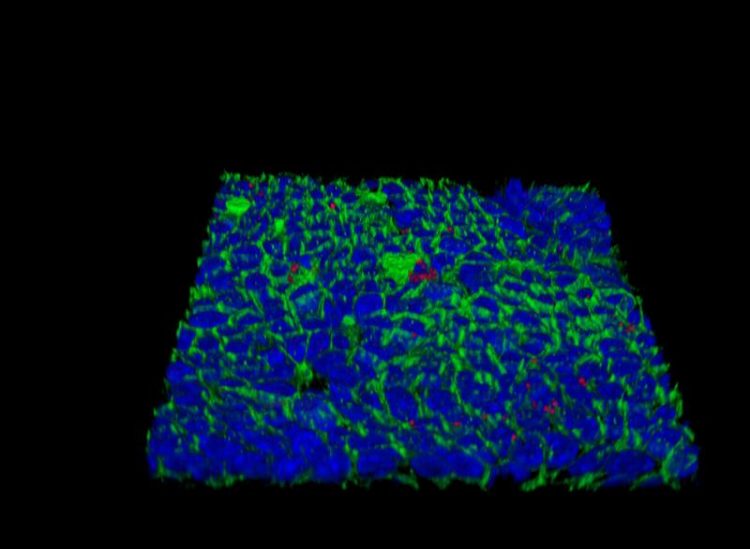
Surface of a Helicobacter-infected mucosoid culture made of normal epithelial cells from the human stomach. Red: Helicobacter pylori, blue: nuclei, green: cell membranes MPI for Infection Biology
While gastritis and gastric ulcer disease used to be put down to stress and dietary factors, it was discovered in the 1980s that the actual culprit is infection with a bacterium, H. pylori. This pathogen is now classed as a type I carcinogen by the WHO, as it is the major risk factor for development of gastric carcinoma.
Attempts to develop a vaccine against H. pylori have been unsuccessful and the new results provide the first potential explanation for these failures: H. pylori blocks the signal that would allow infected cells to summon help from the immune system.
Driven by their interest in understanding the mechanisms by which bacterial infections can give rise to cancer, the laboratory of Prof. Thomas F. Meyer already discovered over a decade ago that H. pylori expresses an enzyme called cholesterol-α-glucosyltransferase to extract the cholesterol it needs from host cells.
Without this enzyme, the bacteria are unable to infect mice. At the time, the group also noted that excess cholesterol in the diet of the mice enhanced clearance of the pathogen but exacerbated inflammation. Exactly how the ability to extract cholesterol was linked to immune evasion remained unclear at the time.
Now, new technologies for growing primary stomach cells in vitro have been instrumental for solving this puzzle. Meyer’s laboratory succeeded in reconstituting the mucosal epithelium of the stomach, yielding so-called “mucosoids” in which gastric cells grow as monolayers with an outer and an inner surface and even produce the mucus that lines and protects the stomach. As a result, the infected “mini-organs” could be kept alive for months.
Infected gastric epithelial cells normally summon immune cells and generate antimicrobial factors that kill the bacteria. This response depends on a crucial immune signaling factor produced by epithelial cells: interferon-γ. Using the mucosoid model, the group found that the bacteria survive as small micro-colonies on top of cells.
This turned out to be due to the absence of so-called “lipid rafts” in the cell membranes, which are required for assembly of interferon receptor complexes. Without these, host cells simply did not respond to interferon-γ at all. The lipid rafts are made of cholesterol, and their destruction is mediated by the bacterium’s cholesterol-α-glucosyltransferase.
“Showing how H. pylori persists in the midst of an acute inflammation explains a whole series of observations that have puzzled researchers for decades and has caused us to think of the infection scenario in an entirely new light” says Dr. Pau Morey, first author of the study. Dr. Francesco Boccellato, who developed the new in vitro model adds: “the mucosoids enable long-term, stable infections to be investigated – and will be an invaluable tool for examining the mechanisms underlying mucosal infections, the mucosal defence and the emergence of cancer.”
Original Publications:
Helicobacter pylori Depletes Cholesterol in Gastric Glands to Prevent Interferon gamma Signaling and Escape the Inflammatory Response.
Morey P, Pfannkuch L, Pang E, Boccellato F, Sigal M, Imai-Matsushima A, Dyer V, Koch M, Mollenkopf H-J, Schlaermann P, Meyer TF (2017). Gastroenterology doi: 10.1053/j.gastro.2017.12.008
Polarised epithelial monolayers of the gastric mucosa reveal insights into mucosal homeostasis and defence against infection.
Boccellato F, Woelffling S, Imai-Matsushima A, Sanchez A, Goosmann C, Schmid M, Berger H, Morey P, Denecke C, Ordemann J, Meyer TF (2018) Gut doi: 10.1136/gutjnl-2017-314540
https://doi.org/10.1053/j.gastro.2017.12.008 Helicobacter pylori Depletes Cholesterol in Gastric Glands to Prevent Interferon gamma Signaling and Escape the Inflammatory Response.
http://dx.doi.org/10.1136/gutjnl-2017-314540 Polarised epithelial monolayers of the gastric mucosa reveal insights into mucosal homeostasis and defence against infection.
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

A universal framework for spatial biology
SpatialData is a freely accessible tool to unify and integrate data from different omics technologies accounting for spatial information, which can provide holistic insights into health and disease. Biological processes…

How complex biological processes arise
A $20 million grant from the U.S. National Science Foundation (NSF) will support the establishment and operation of the National Synthesis Center for Emergence in the Molecular and Cellular Sciences (NCEMS) at…

Airborne single-photon lidar system achieves high-resolution 3D imaging
Compact, low-power system opens doors for photon-efficient drone and satellite-based environmental monitoring and mapping. Researchers have developed a compact and lightweight single-photon airborne lidar system that can acquire high-resolution 3D…





















