Chemists Discover Way Nose Perceives Common Class of Odors
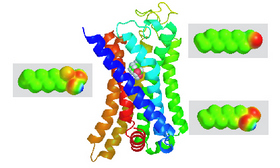
Image shows the mouse aldehyde receptor OR-I7 as a homology model with the gem-diol form of octanal. The chemical structures surrounding the receptor depict some of those used in the study.
According to a team led by CCNY Associate Professor of Chemistry Kevin Ryan and Columbia biologist Stuart Firestein, some of the nose's many aldehyde receptors don't detect the aldehyde by its structure and shape directly. Rather, the aldehyde is recognized by its ability to undergo a chemical reaction with water, likely after entering the nose.
Odorant receptors make up a large family of cell membrane proteins that monitor inhaled air on neurons within the nose. Aldehydes, meanwhile, are found in a variety of natural sources like herbs, flowers and fruit. They are typically fresh-smelling chemicals, and synthetic aldehydes are important to the flavor and fragrance industry.
“Once exposed to air, aldehydes have a limited lifetime as oxygen slowly converts them into less savory, even malodorous chemicals,” said Professor Ryan. “It's not surprising then that the nose is adept at detecting aldehydes, and distinguishing them from structurally similar chemical groups.”
His team found that for some receptors it's the aldehyde's chemical reactivity, not its inherent shape, that tells the nose there are aldehydes in the air.
In our experiments, some of the many odorant receptors that detected the eight-carbon aldehyde octanal recognized the aldehyde portion of the molecule by its ability to morph into a completely different chemical group, known as a gem-diol,” he added. “Since this reaction is unique to aldehydes, it serves as a means to discriminate them from similarly shaped chemical groups.”
Researchers from Duke University and Hebrew University in Israel were also involved in the study. The research at CCNY was funded by U.S. Army Research Laboratory and Office. The results will be published in the journal “ACS Chemical Biology.”
About The City College of New York
Since 1847, The City College of New York has provided low-cost, high-quality education for New Yorkers in a wide variety of disciplines. More than 16,000 students pursue undergraduate and graduate degrees in: the College of Liberal Arts and Sciences; the Bernard and Anne Spitzer School of Architecture; the School of Education; the Grove School of Engineering; the Sophie Davis School of Biomedical Education, and the Colin Powell School for Civic and Global Leadership. U.S. News, Princeton Review and Forbes all rank City College among the best colleges and universities in the United States.
Jay Mwamba
p: 212.650.7580
e:jmwamba@ccny.cuny.edu
Media Contact
More Information:
http://www.ccny.cuny.edu/news/distinguishing-orders.cfmAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

A universal framework for spatial biology
SpatialData is a freely accessible tool to unify and integrate data from different omics technologies accounting for spatial information, which can provide holistic insights into health and disease. Biological processes…

How complex biological processes arise
A $20 million grant from the U.S. National Science Foundation (NSF) will support the establishment and operation of the National Synthesis Center for Emergence in the Molecular and Cellular Sciences (NCEMS) at…

Airborne single-photon lidar system achieves high-resolution 3D imaging
Compact, low-power system opens doors for photon-efficient drone and satellite-based environmental monitoring and mapping. Researchers have developed a compact and lightweight single-photon airborne lidar system that can acquire high-resolution 3D…





















