A Freiburg research team deciphers how stem cells decide their identity
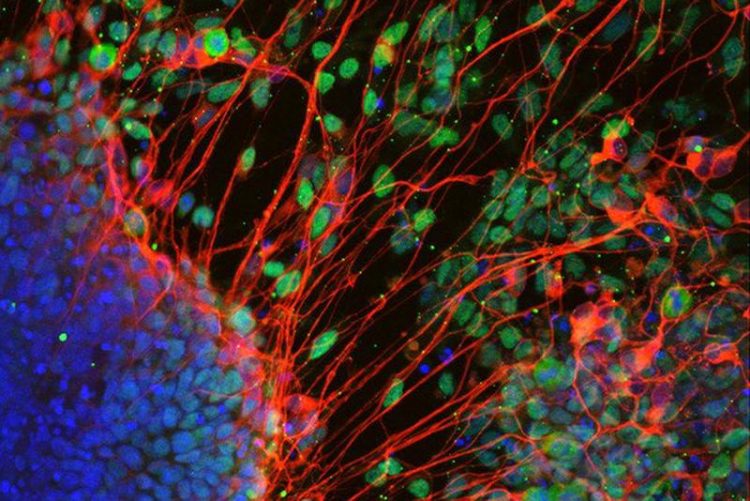
Neurons derived from stem cell in the absence of T-Box factors. Source: Carsten Schwan/ Jelena Tosic
This is achieved at least partially through selective usage of the genes for each different cell type, despite the presence of the identical genetic information in every cell in the body.
The scientists have published their findings in the journal “Nature Cell Biology”.
The undifferentiated stem cells of the embryo develop either into cells of the nervous system, the so-called neuroectoderm, or into cells of the meso- and endoderm, from which, for example, many different cell types of the internal organs or the muscles develop.
For over 25 years it has been known that this decision is regulated by embryonic signaling molecules, such as TGFβ and Wnt signals. So far, however, it has remained unclear exactly how these signals control this first decision of cell differentiation.
The study, carried out in the context of Tosic's doctoral thesis, shows that the embryonic TGFβ and Wnt signals are transmitted by gene-regulating transcription factors of the T-box factor family, namely Eomes and Brachyury. These factors are responsible for “turning on” the differentiation gene programs for all meso- and endoderm cells.
At the same time, these T-box factors also act as gene repressors, preventing the formation of neural tissue by suppressing the corresponding gene programs. This involves changes in the structure but not the content of the genetic information in the cell nucleus.
“The results of the study represent a crucial step towards understanding the basic mechanisms of how cells develop their future identity during development,” says Arnold. They also allow further studies on how cell identity is permanently encoded in a cell.
Sebastian Arnold works at the Institute for Experimental and Clinical Pharmacology and Toxicology of the University of Freiburg’s Faculty of Medicine. He is also involved in the Collaborative Research Centres 850, 1140, and 994 as well as the Freiburg Cluster of Excellence CIBSS – Centre for Integrated Biological Signalling Studies.
Original publication:
Tosic, J., Kim, G.-J., Pavlovic, M., Schröder, C.M., Mersiowsky, S.-L., Barg, M., Hofherr, A., Probst, S., Köttgen, M., Hein, L., and Arnold, S.J. (2019): Eomes and Brachyury control pluripotency exit and germ layer segregation by changing the chromatin state. In: Nature Cell Biol. DOI: http://dx.doi.org/10.1038/s41556-019-0423-1
Contact:
Prof. Dr. Sebastian Arnold
Research Group for Regenerative Pharmacology
Institute of Experimental & Clinical Pharmacology and Toxicology
University of Freiburg
Phone: 0761 / 203-96819
sebastian.arnold@pharmakol.uni-freiburg.de
https://www.pr.uni-freiburg.de/pm-en/press-releases-2019/cellular-identity?set_l…
Media Contact
More Information:
http://www.uni-freiburg.de/All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

High-energy-density aqueous battery based on halogen multi-electron transfer
Traditional non-aqueous lithium-ion batteries have a high energy density, but their safety is compromised due to the flammable organic electrolytes they utilize. Aqueous batteries use water as the solvent for…

First-ever combined heart pump and pig kidney transplant
…gives new hope to patient with terminal illness. Surgeons at NYU Langone Health performed the first-ever combined mechanical heart pump and gene-edited pig kidney transplant surgery in a 54-year-old woman…

Biophysics: Testing how well biomarkers work
LMU researchers have developed a method to determine how reliably target proteins can be labeled using super-resolution fluorescence microscopy. Modern microscopy techniques make it possible to examine the inner workings…





















