Some aggressive cancers may respond to anti-inflammatory drugs
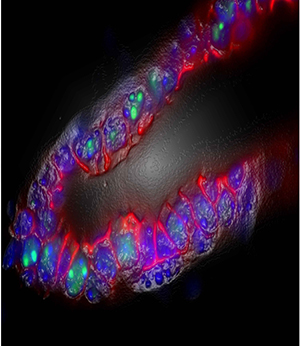
Raleigh Kladney A mouse mammary gland missing the tumor suppressor p53 shows expression of ARF (green), now known for a backup role in protecting cells from becoming cancerous. If both p53 and ARF are mutated, the tumors that form are aggressive and may benefit from treatment with anti-inflammatory drugs called JAK inhibitors, currently prescribed for rheumatoid arthritis.
Studying triple-negative breast cancer, researchers at Washington University School of Medicine in St. Louis found that some aggressive tumors rely on an antiviral pathway that appears to drive inflammation, widely recognized for roles in cancer, rheumatoid arthritis and other inflammatory diseases.
The tumors that activate this particular antiviral pathway always have dysfunctional forms of the proteins p53 and ARF, both encoded by genes known for being highly mutated in various cancers. The investigators found that the two genes compensate for each other. If both are mutated, the tumors that form are more aggressive than if only one of these genes is lost.
When both genes are lost and the antiviral pathway is activated, patients may benefit from a class of anti-inflammatory drugs called JAK inhibitors, currently prescribed for rheumatoid arthritis.
The investigators report their findings in a recent issue of the journal Cell Reports.
Until now, even though ARF was known to be expressed in some tumors with mutated p53, ARF largely was thought to be nonfunctional in this scenario. But the investigators showed that in the absence of p53, ARF actually protects against even more aggressive tumor formation.
“It’s probably inaccurate to say that ARF completely replaces p53, which is a robust tumor suppressor with multiple ways of working,” said senior author Jason D. Weber, PhD, associate professor of medicine. “But it appears the cell has set up a sort of backup system with ARF. It’s not surprising that these are the two most highly mutated tumor suppressors in cancer. Because they’re backing one another up, the most aggressive tumors form when you lose both.”
Weber and his colleagues studied triple-negative breast cancer because these tumors often show mutations in both p53 and ARF. Triple-negative breast tumors are treated with surgery, chemotherapy and radiation since targeted therapies commonly used against hormone-driven breast cancers are not effective.
In a finding Weber called surprising, the researchers showed that most triple-negative tumors lacking p53 and ARF turn on a pathway involved in the innate immune response to viral infection.
“It’s not the level of activation you would see in a true antiviral response, but it’s higher than normal,” Weber said. “We are interested in studying whether this antiviral response is creating a local environment of inflammation that supports more aggressive tumors.”
Weber and his colleagues knew that a signaling protein family known as JAK is upstream of the antiviral pathway they showed to be driving tumor growth.
“There are JAK inhibitors in use for rheumatoid arthritis and being tested against a number of other conditions,” Weber said. “Our data suggest that these anti-inflammatory drugs may be a way to treat some patients missing both p53 and ARF.”
The drugs potentially could benefit patients in whom both genes are lost, Weber added. If either p53 or ARF is present, this antiviral pathway is not active and therefore not playing a role in driving tumor growth.
Weber and his team are collaborating with specialists in lung, breast and pancreatic cancer to identify patients with mutations in both genes and to find out whether such patients might benefit from JAK inhibitors.
This work was supported by the National Institutes of Health (NIH), the National Cancer Institute (NCI) of the NIH, a Clinical and Translational Science Award (CTSA) from the NIH and a Department of Defense Era of Hope Scholar grant. Grant numbers R01CA120436, 1K12CA167540, UL1RR024992, and 5T32GM007067.
Forys JT, Kuzmicki CE, Saporita AJ, Winkeler CL, Maggi Jr. LB, Weber JD. ARF and p53 coordinate tumor suppression of an oncogenic IFN-beta-STAT1-ISG15 signaling axis. Cell Reports. April 2014.
Washington University School of Medicine’s 2,100 employed and volunteer faculty physicians also are the medical staff of Barnes-Jewish and St. Louis Children’s hospitals. The School of Medicine is one of the leading medical research, teaching and patient-care institutions in the nation, currently ranked sixth in the nation by U.S. News & World Report. Through its affiliations with Barnes-Jewish and St. Louis Children’s hospitals, the School of Medicine is linked to BJC HealthCare.
Media Contact
More Information:
https://news.wustl.edu/news/Pages/27051.aspxAll latest news from the category: Health and Medicine
This subject area encompasses research and studies in the field of human medicine.
Among the wide-ranging list of topics covered here are anesthesiology, anatomy, surgery, human genetics, hygiene and environmental medicine, internal medicine, neurology, pharmacology, physiology, urology and dental medicine.
Newest articles

Microscopic basis of a new form of quantum magnetism
Not all magnets are the same. When we think of magnetism, we often think of magnets that stick to a refrigerator’s door. For these types of magnets, the electronic interactions…

An epigenome editing toolkit to dissect the mechanisms of gene regulation
A study from the Hackett group at EMBL Rome led to the development of a powerful epigenetic editing technology, which unlocks the ability to precisely program chromatin modifications. Understanding how…

NASA selects UF mission to better track the Earth’s water and ice
NASA has selected a team of University of Florida aerospace engineers to pursue a groundbreaking $12 million mission aimed at improving the way we track changes in Earth’s structures, such…





















