Researchers Discover Possible New Target to Attack Flu Virus
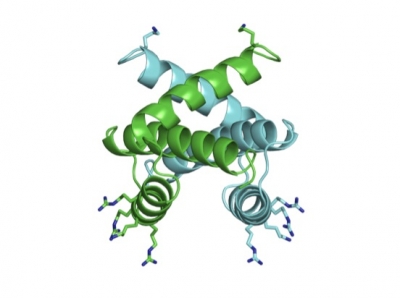
This region of the NS1 viral protein binds the host protein DDX21, making it a potential target for new antivirals against the influenza virus
Better antiviral drugs could help the millions of people annually infected by flu, which kills up to 500,000 people each year.
When an influenza virus infects a human cell, it uses some of the host's cellular machinery to make copies of itself, or replicate. In this study, the researchers discovered that a protein produced by human body cells, DDX21, blocks this replication process. They also discovered that a protein created by the virus, NS1, in turn blocks DDX21 and promotes viral replication.
“If you could figure out how to stop NS1 from binding to DDX21, you could stop the virus cold,” said Robert Krug, a professor in the College of Natural Sciences at The University of Texas at Austin and corresponding author on the study, which appears today in the journal Cell Host and Microbe.
Krug said that in addition to countering the body's defense mechanisms, the viral NS1 protein actually performs other important roles for the virus, such as inhibiting the host's synthesis of interferon, a key antiviral protein.
“It means that if you could block that NS1 function, you'd be blocking not only its interaction with DDX21 but many other important functions, so it's a great target,” said Krug.
The need for new antiviral drugs against the influenza virus is great. Because flu vaccines are not 100 percent effective, antiviral drugs play an important role in fast-spreading epidemics. Yet influenza A viruses are developing resistance to antiviral drugs currently in use.
Krug and his team discovered that the viral NS1 protein is often associated, or bound together, with the host DDX21 protein in infected human body cells. To understand what role DDX21 might play in virus replication, the researchers used a technique called siRNA gene silencing to knock down the production of DDX21 in infected cells. When they did, virus replication increased 30 fold.
“That told us that DDX21 is a host restriction factor, that it inhibits replication,” said Krug. “That was the key to understanding what was happening. It was an exciting moment.”
Next, the researchers discovered that DDX21 blocks replication by binding to a protein that the virus needs to replicate, called PB1. Finally, they discovered that NS1 binds to DDX21 and makes PB1 available again for replication. This result confirmed that NS1 was indeed the countermeasure used by the virus to get around the body's natural defense mechanism.
Krug's co-authors are Guifang Chen, Chien-Hung Liu and Ligang Zhou, all from The University of Texas at Austin.
Support for this research was provided by a grant from the National Institutes of Health.
Media Contact
More Information:
http://cns.utexas.edu/news/possible-new-target-to-attack-flu-virusAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

A universal framework for spatial biology
SpatialData is a freely accessible tool to unify and integrate data from different omics technologies accounting for spatial information, which can provide holistic insights into health and disease. Biological processes…

How complex biological processes arise
A $20 million grant from the U.S. National Science Foundation (NSF) will support the establishment and operation of the National Synthesis Center for Emergence in the Molecular and Cellular Sciences (NCEMS) at…

Airborne single-photon lidar system achieves high-resolution 3D imaging
Compact, low-power system opens doors for photon-efficient drone and satellite-based environmental monitoring and mapping. Researchers have developed a compact and lightweight single-photon airborne lidar system that can acquire high-resolution 3D…





















