New jigsaw piece for the repair of DNA crosslinks
Crosslink-inducing agents used in chemotherapy. pictures:UZH
UZH researchers have now discovered another piece in the puzzle for the removal of extremely dangerous DNA lesions. Faithful and efficient repair of so-called crosslinks requires a collaboration between a specific signalling and repair protein. As crosslink-inducing agents are used in chemotherapy, the new insights are also important for the development of better anti-cancer treatment strategies.
Environmental influences such as ionizing radiation, intense heat or various chemical substances damage the DNA constantly. Only thanks to efficient repair systems can mutations – changes in the DNA – largely be prevented. DNA crosslinks that covalently link both strands of the DNA double helix are among the most dangerous DNA lesions. Crosslinks block DNA replication and can thus cause cell death.
Moreover, their faulty repair can trigger the development of tumors. Crosslink repair is highly complex and only vaguely understood today. A team of cancer researchers headed by Alessandro Sartori from the University of Zurich now reveals interesting details as to how cells recognize crosslink damage. In their study recently published in Cell Reports, the scientists demonstrate that the interplay between two specific proteins is crucial for the flawless repair of crosslink damage.
Repair protein recognizes crosslink damage with the aid of a signal protein
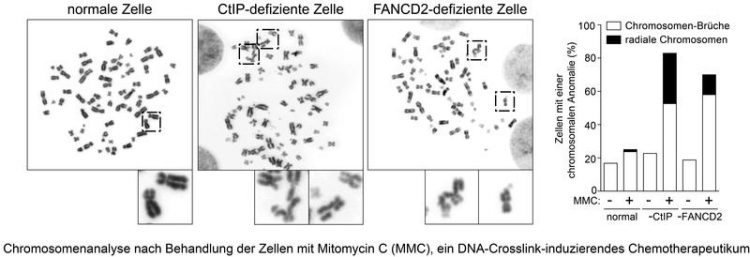
For their study, the researchers examined the Fanconi anemia signal pathway, which coordinates the complex repair of crosslinks, with the aid of genetically modified and unchanged cells. Sartori and his team wanted to find out whether and how the signal pathway and the repair protein CtIP interact with one another. “We are able to show that CtIP recognizes and repairs crosslinks efficiently with the aid of the Fanconi anemia signal pathway, or FANCD2 to be more precise,” explains Sartori.
The scientists also discovered the point where CtIP attaches itself to the FANCD2 protein. According to the researchers, the interplay between the two proteins is necessary for the flawless and smooth repair of crosslink damage as it prevents the relocation of entire chromosome sections to another position (see figure). Referred to as chromosomal translocation, the process is one of the main causes of the development of cancer.
These days, substances that specifically trigger crosslink damage are used in cancer chemotherapy. The new findings are therefore important for both our understanding of the development of cancer and the further development of improved drugs.
Literature:
Olga Murina, Christine von Aesch, Ufuk Karakus, Lorenza P. Ferretti, Hella A. Bolck, Kay Hänggi, and Alessandro A. Sartori. FANCD2 and CtIP Cooperate to Repair DNA Interstrand Crosslinks. Cell Reports (2014). May 1, 2014. http://dx.doi.org/10.1016/j.celrep.2014.03.069
Fanconi anemia
Fanconi anemia (FA) is a rare congenital disorder that was first described in 1927 by Guido Fanconi (1892–1979), Professor of Pediatrics at the University of Zurich. Fanconi anemia is triggered by mutations in genes that regulate the repair of DNA crosslinks. Patients who suffer from Fanconi anemia display bone marrow failure already during childhood and have a risk of developing cancer that is about 1,000 times higher compared to healthy individuals. Only around a third of Fanconi anemia patients live beyond the age of 30.
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

A universal framework for spatial biology
SpatialData is a freely accessible tool to unify and integrate data from different omics technologies accounting for spatial information, which can provide holistic insights into health and disease. Biological processes…

How complex biological processes arise
A $20 million grant from the U.S. National Science Foundation (NSF) will support the establishment and operation of the National Synthesis Center for Emergence in the Molecular and Cellular Sciences (NCEMS) at…

Airborne single-photon lidar system achieves high-resolution 3D imaging
Compact, low-power system opens doors for photon-efficient drone and satellite-based environmental monitoring and mapping. Researchers have developed a compact and lightweight single-photon airborne lidar system that can acquire high-resolution 3D…





















