Making the Good Much Better Against Cancer
Tubulin, the building-block protein of cellular microtubules, is a well-validated cancer drug target: Disrupting tubulin polymerization affects cytoskeletal function and thus cell division. The vinca alkaloids, taxanes, and other taxane site binders have found wide therapeutic application and constitute one of the most successful classes of anticancer agent.
Nevertheless, current drugs that target tubulin still suffer from limited therapeutic window, acquired resistance, lack of oral bioavailability, and problematic formulation.
As reported in ChemMedChem, Barry Potter’s research group at the University of Bath (UK), along with colleagues at the NCI (USA), Ipsen (France), and Imperial College London, focused on a natural steroidal lead for introducing a new set of modifications that confer good oral activity and delivery and that impart resistance to metabolism.
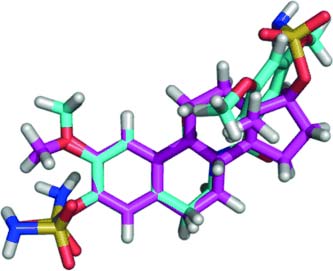
“Two compounds showed in vitro anti-angiogenic activity, and X-ray analysis of one reveals that electrostatic repulsion between two adjacent carbonyl groups […] dictates adoption of a 'steroid-like' conformation that might partially explain the excellent in vitro activities,” says Potter. Indeed, the most active compounds are nearly equivalent to another compound currently in clinical trials as inhibitors of tubulin assembly, and some were shown to be anti-angiogenic. Moreover, the compounds are easily synthesized and amenable to aqueous formulation.
About the Author
Professor Barry Potter holds the established chair of Medicinal Chemistry at the University of Bath and is also Visiting Professor of Medicinal Chemistry at Oxford University. His research interests are in mechanistic enzymology, the chemistry of signal transduction, and anticancer drug design and discovery, and he has brought academically discovered drugs to multiple clinical trials. He is a Fellow of the Royal Society of Chemistry (RSC) and the Society of Biology, was elected to the UK National Academy of Medical Sciences, has won four interdisciplinary RSC medals, the GlaxoSmithKline International Achievement Award, and was named Investigator of the Year at the 2012 European Life Science Awards.
Author: Barry V. L. Potter, University of Bath (UK), http://www.bath.ac.uk/pharmacy/contacts/academics/barry_potter/
Title: Synthesis, Anti-tubulin and Antiproliferative SAR of Steroidomimetic Dihydroisoquinolinones
ChemMedChem, Permalink to the article: http://dx.doi.org/10.1002/cmdc.201400017
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

A universal framework for spatial biology
SpatialData is a freely accessible tool to unify and integrate data from different omics technologies accounting for spatial information, which can provide holistic insights into health and disease. Biological processes…

How complex biological processes arise
A $20 million grant from the U.S. National Science Foundation (NSF) will support the establishment and operation of the National Synthesis Center for Emergence in the Molecular and Cellular Sciences (NCEMS) at…

Airborne single-photon lidar system achieves high-resolution 3D imaging
Compact, low-power system opens doors for photon-efficient drone and satellite-based environmental monitoring and mapping. Researchers have developed a compact and lightweight single-photon airborne lidar system that can acquire high-resolution 3D…





















