Image or Mirror Image?
It is not always easy to distinguish between images and mirror images of molecules, but this knowledge is important when one image of a molecule is a drug and the mirror image is toxic. One new approach to this may be chiral recognition in the gas phase.
This involves using synchrotron radiation (highly energetic photons from a particle accelerator) to eject electrons from the molecules and analyzing their trajectories. In the journal Angewandte Chemie, German researchers have now demonstrated that such experiments also work with a compact laser system.
The trick is to replace the individual high-energy photon with three laser photons that excite the molecule through intermediate levels until it releases an electron (this method is known as REMPI, Resonance-Enhanced Multi-Photon Ionization). “It is thus possible to eject electrons with less energetic but more intense light,” explains Thomas Baumert of the University of Kassel.
For the measurements, the light must be circularly polarized. What does this mean? “Ordinary” light consists of waves that oscillate in all spatial directions perpendicular to their direction of travel. If light is linearly polarized, the light waves oscillate exclusively in one plane. When light is circularly polarized, the light wave oscillates in a helical form, because its amplitude describes a circle around the axis of travel – either to the right or the left.
Molecules in the gas phase are randomly oriented and thus encounter the laser light from all possible angles; the ejected electrons also fly off in every possible direction as they leave the molecule. By using both a special configuration for measurement and special calculation processes, the team is able to determine the distribution of the angles of the electrons’ flight paths. In the case of linearly polarized light, the distribution is symmetrical.
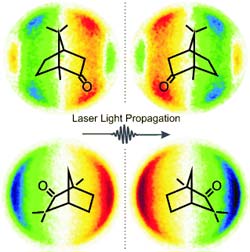
“However, when the electrons are ejected by circularly polarized light, we find a distinct asymmetry to the angles at which the free electrons are found in relation to the laser beam,” reports Baumert. “This asymmetry is inverted if left circularly polarized light is used instead of right, an effect known as photoelectron circular dichroism. We observe the same effect when we keep the circular polarization the same but change from the “right handed” to the “left handed” structure of the chiral molecule being observed.”
The researchers were able to demonstrate this with the chiral compounds camphor and fenchone.
“This circular dichroism effect has previously only been observed with synchrotron radiation. In contrast, our procedure uses a compact laser system, so that this method is not limited to basic laboratory research but, because of the magnitude of the observed effects, may also find its way into analysis,” according to Baumert.
About the Author
Dr. Thomas Baumert has been Professor of Experimental Physics at the University of Kassel for over a decade. His research interests include femtosecond spectroscopy and ultrasound control of matter by means of tailored light fields.
Author: Thomas Baumert, Universität Kassel (Germany), http://www.physik.uni-kassel.de/de/484.html
Title: Circular Dichroism in the Photoelectron Angular Distributions of Camphor and Fenchone from Multiphoton Ionization with Femtosecond Laser Pulses
Angewandte Chemie International Edition, Permalink to the article: http://dx.doi.org/10.1002/anie.201109035
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

High-energy-density aqueous battery based on halogen multi-electron transfer
Traditional non-aqueous lithium-ion batteries have a high energy density, but their safety is compromised due to the flammable organic electrolytes they utilize. Aqueous batteries use water as the solvent for…

First-ever combined heart pump and pig kidney transplant
…gives new hope to patient with terminal illness. Surgeons at NYU Langone Health performed the first-ever combined mechanical heart pump and gene-edited pig kidney transplant surgery in a 54-year-old woman…

Biophysics: Testing how well biomarkers work
LMU researchers have developed a method to determine how reliably target proteins can be labeled using super-resolution fluorescence microscopy. Modern microscopy techniques make it possible to examine the inner workings…





















