Novel NIST process is a low-cost route to ultrathin platinum films
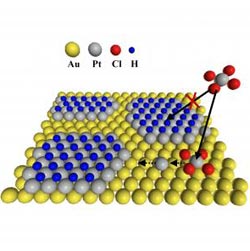
Schematic shows self-quenched platinum deposition on a gold surface. Under a high driving voltage, platinum in solution (bound to four chloride atoms) can shed the chloride and bind to a location on the gold. Hydrogen rapidly adsorbs on the platinum, ensuring that the platinum forms an even surface a single atom thick.<br><br>Credit: Gokcen/NIST<br>
The new process exploits an unexpected feature of electrodeposition of platinum—if you drive the reaction much more strongly than usual, a new reaction steps in to shuts down the metal deposition process, allowing an unprecedented level of control of the film thickness.
Platinum is a widely used industrial catalyst—in automobile catalytic converters and hydrogen fuel cells—as well as a key component in microelectronics, so the discovery may have widespread application in the design and manufacture of platinum-based devices.
The metal is rare, and hence very pricey, so materials engineers try to use it sparingly as a thin layer on a substrate. They'd like to be able to control the deposition process down to uniform, single layers of atoms. Unfortunately, platinum doesn't always cooperate.
The model system studied at NIST—depositing a platinum layer on gold by electroplating—demonstrates the challenging nature of the problem. A voltage is applied to drive the deposition of platinum from an electrode onto the gold surface in an aqueous solution. Normally, this leads to a patchy and rough surface rather than the desired smooth and even layer of platinum, because platinum tends to attach first to any defects on the gold surface, and then tends to attach to itself, rather than the gold.
The NIST team has found that increasing the voltage, the driving force of the reaction, far higher than normal to the point where the water molecules start to break down and hydrogen ions form, leads to an unexpected and useful result. The hydrogen quickly forms a layer covering the freshly deposited platinum islands and completely quenches further metal deposition. Using a battery of analytic techniques, including a quartz crystal microbalance, X-ray photoelectron spectroscopy and scanning tunneling microscopy, the group found that the formation of the hydrogen layer was rapid enough to restrict deposition to the formation of a single layer of platinum atoms. The team further discovered that by pulsing the applied voltage, they could selectively remove the hydrogen layer to enable the platinum deposition process to be repeated to form another layer.
The deposition process occurs in a single plating bath and is surprisingly fast—1,000 times faster than making comparable films using molecular beam epitaxy, for example. It's also faster, simpler and less prone to contamination than other electrochemical techniques for depositing platinum films, making it much less expensive.
The novel technique, the researchers say, may also work with a number of other metal and alloy combinations, a subject of ongoing research.
* Y. Liu, D. Gokcen, U. Bertocci and T.P. Moffat. Self-terminating growth of platinum films by electrochemical deposition. Science, v. 338, 1327, Dec. 7, 2012. Doi: 10.1126/science.1228925.
Media Contact
More Information:
http://www.nist.govAll latest news from the category: Materials Sciences
Materials management deals with the research, development, manufacturing and processing of raw and industrial materials. Key aspects here are biological and medical issues, which play an increasingly important role in this field.
innovations-report offers in-depth articles related to the development and application of materials and the structure and properties of new materials.
Newest articles

High-energy-density aqueous battery based on halogen multi-electron transfer
Traditional non-aqueous lithium-ion batteries have a high energy density, but their safety is compromised due to the flammable organic electrolytes they utilize. Aqueous batteries use water as the solvent for…

First-ever combined heart pump and pig kidney transplant
…gives new hope to patient with terminal illness. Surgeons at NYU Langone Health performed the first-ever combined mechanical heart pump and gene-edited pig kidney transplant surgery in a 54-year-old woman…

Biophysics: Testing how well biomarkers work
LMU researchers have developed a method to determine how reliably target proteins can be labeled using super-resolution fluorescence microscopy. Modern microscopy techniques make it possible to examine the inner workings…





















