Organic electronics — how to make contact between carbon compounds and metal
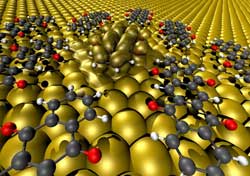
Upon contact between the oxygen atoms protruding from the backbone and the metal, the molecules' internal structure changed in such a way that they lost their semiconducting properties and instead adopted the metallic properties of the surface.<br><br>Credit: Visualisation: Georg Heimel/HU Berlin<br>
Now, an international team of scientists around Dr. Georg Heimel and Prof. Norbert Koch from the HZB and the Humboldt University Berlin has unraveled the mystery of what these molecules have in common. Their discovery enables more focused improvements to contact layers between metal electrodes and active materials in organic electronic devices.
“We have been working on this question for a number of years now and could at last come up with a conclusive picture using a combination of several experimental methods and theoretical calculations,” Georg Heimel explains. The researchers systematically examined different types of molecules whose backbones consist of the same chain of fused aromatic carbon rings. They differed in just one little detail: the number of oxygen atoms projecting from the backbone. These modified molecules were placed on the typical contact metals gold, silver, and copper.
Using photoelectron spectroscopy (UPS and XPS) at HZB's own BESSY II synchrotron radiation source, the researchers were able to identify chemical bonds that formed between the metal surfaces and the molecules as well as to measure the energy levels of the conduction electrons. Colleagues from Germany's Tübingen University determined the exact distance between the molecules and the metal surfaces using x-ray standing wave measurements taken at the ESRF synchrotron radiation source in Grenoble, France.
These experiments showed that, upon contact between the oxygen atoms protruding from the backbone and several of the metals, the molecules' internal structure changed in such a way that they lost their semiconducting properties and instead adopted the metallic properties of the surface. Despite similar prerequisites, this effect was not observed for the “bare”-backbone molecule. From the observation which molecules underwent these kinds of drastic changes on what metal, the researchers could derive general guidelines. “At this point, we have a pretty good sense of how molecules ought to look like and what their properties should be if they are to be good mediators between active organic materials and metal contacts, or, as we like to call it, good at forming soft metallic contacts,” says Heimel.
Experts from a number of other German universities and from research facilities in Suzhou (China), Iwate and Chiba (Japan), and ESRF (France) have also contributed substantially to this publication.
For additional information please contact Prof. Dr. Norbert Koch
Fon: +49 30-20 93 78 19
E-Mail: norbert.koch@helmholtz-berlin.de
E-Mail: norbert.koch@physik.hu-berlin.de
Media Contact
More Information:
http://www.hu-berlin.deAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Combining robotics and ChatGPT
TUM professor uses ChatGPT for choreographies with flying robots. Prof. Angela Schoellig has proved that large language models can be used safely in robotics. ChatGPT develops choreographies for up to…

How the Immune System Learns from Harmless Particles
Our lungs are bombarded by all manner of different particles every single day. Whilst some are perfectly safe for us, others—known as pathogens—have the potential to make us ill. The…

Biomarkers identified for successful treatment of bone marrow tumours
CAR T cell therapy has proven effective in treating various haematological cancers. However, not all patients respond equally well to treatment. In a recent clinical study, researchers from the University…