Stickier than expected: Hydrogen binds to graphene in 10 femtoseconds
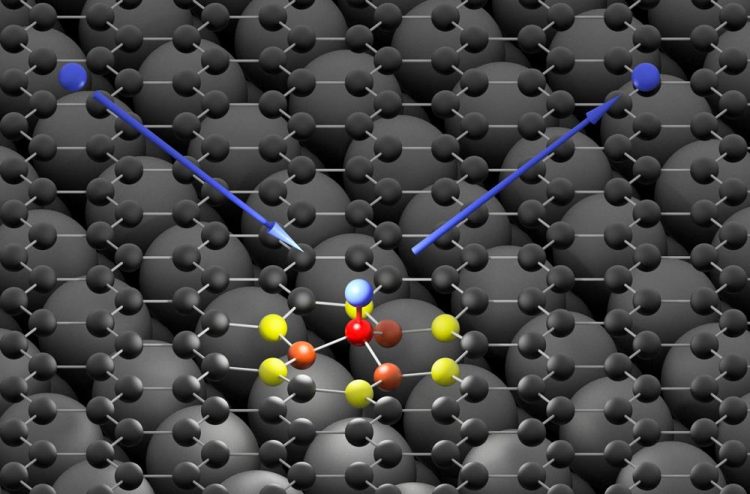
The hydrogen atom (blue) hits the graphene surface (black) and forms an ultra-fast bond with a carbon atom (red). The high energy of the impinging hydrogen atom is first absorbed by neighboring carbon atoms (orange and yellow) and then passed on to the graphene surface in form of a sound wave Credit: Oliver Bünermann / Max Planck Institute for Biophysical Chemistry & University of Göttingen
Graphene is celebrated as an extraordinary material. It consists of pure carbon, only a single atomic layer thick. Nevertheless, it is extremely stable, strong, and even conductive. For electronics, however, graphene still has crucial disadvantages.
It cannot be used as a semiconductor, since it has no bandgap. By sticking hydrogen atoms to graphene such a bandgap can be formed. Now researchers from Göttingen and Pasadena (USA) have produced an “atomic scale movie” showing how hydrogen atoms chemically bind to graphene in one of the fastest reactions ever studied. (Science, April 25, 2019)
The international research team bombarded graphene with hydrogen atoms. “The hydrogen atom behaved quite differently than we expected,” says Alec Wodtke, head of the Department of Dynamics at Surfaces at the Max Planck Institute (MPI) for Biophysical Chemistry and professor at the Institute of Physical Chemistry at the University of Göttingen.
“Instead of immediately flying away, the hydrogen atoms 'stick' briefly to the carbon atoms and then bounce off the surface. They form a transient chemical bond,” Wodtke reports.
And something else surprised the scientists: The hydrogen atoms have a lot of energy before they hit the graphene, but not much left when they fly away. Hydrogen atoms lose most of their energy on collision, but where does it go?
To explain these surprising experimental observations, the Göttingen MPI researcher Alexander Kandratsenka, in cooperation with colleagues at the California Institute of Technology, developed theoretical methods, which they simulated on the computer and then compared to their experiments.
With these theoretical simulations, which agree well with the experimental observations, the researchers were able to reproduce the ultra-fast movements of atoms forming the transient chemical bond. “This bond lasts for only about ten femtoseconds – ten quadrillionths of a second. This makes it one of the fastest chemical reactions ever observed directly,” Kandratsenka explains.
“During these ten femtoseconds, the hydrogen atom can transfer almost all its energy to the carbon atoms of the graphene and it triggers a sound wave that propagates outward from the point of the hydrogen atom impact over the graphene surface, much like a stone that falls into water and triggers a wave,” says Kandratsenka.
The sound wave contributes to the fact that the hydrogen atom can bind more easily to the carbon atom than the scientists had expected and previous models had predicted.
The results of the research team provide fundamentally new insights into chemical bonding. In addition, they are of great interest to industry. Sticking Hydrogen atoms to graphene can produce a bandgap, making it a useful semiconductor and much more versatile in electronics.
The effort involved in setting up and running these experiments was enormous, revealed Oliver Bünermann, project group leader at the University of Göttingen. “We had to carry them out in ultra-high vacuum to keep the graphene surface perfectly clean.”
The scientists also had to use a large number of laser systems to prepare the hydrogen atoms before the experiment and to detect them after the collision. According to Bünermann, the excellent technical staff in the workshops at the MPI for Biophysical Chemistry and at the University of Göttingen were essential to the project's success.
###
Further information: http://www.
Original publication: Hongyan Jiang, Marvin Kammler, Feizhi Ding, Yvonne Dorenkamp, Frederick R. Manby, Alec. M. Wodtke, Thomas F. Miller, Alexander Kandratsenka, Oliver Bünermann: Imaging covalent bond formation by H atom scattering from graphene. Science 364, 6438, 379-382, doi: 10.1126/science.aaw6378 (2019).
Contact:
Professor Alec Wodtke,
Group of Dynamics at Surfaces
MPI for Biophysical Chemistry & University of Göttingen
Phone: +49 551 201-1261
Email: alec.wodtke@mpibpc.mpg.de
Media Contact
All latest news from the category: Materials Sciences
Materials management deals with the research, development, manufacturing and processing of raw and industrial materials. Key aspects here are biological and medical issues, which play an increasingly important role in this field.
innovations-report offers in-depth articles related to the development and application of materials and the structure and properties of new materials.
Newest articles

A universal framework for spatial biology
SpatialData is a freely accessible tool to unify and integrate data from different omics technologies accounting for spatial information, which can provide holistic insights into health and disease. Biological processes…

How complex biological processes arise
A $20 million grant from the U.S. National Science Foundation (NSF) will support the establishment and operation of the National Synthesis Center for Emergence in the Molecular and Cellular Sciences (NCEMS) at…

Airborne single-photon lidar system achieves high-resolution 3D imaging
Compact, low-power system opens doors for photon-efficient drone and satellite-based environmental monitoring and mapping. Researchers have developed a compact and lightweight single-photon airborne lidar system that can acquire high-resolution 3D…





















