Researchers Create New Organic Gel Nanomaterials
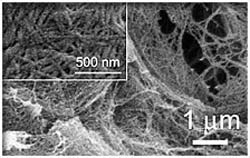
“We are using the building blocks provided by nature to create new nanomaterials that are completely reversible and environmentally benign,” said Jonathan Dordick, the Howard P. Isermann ‘42 Professor of Chemical and Biological Engineering at Rensselaer Polytechnic Institute. “The importance of this finding is the ability to use the same naturally occurring enzyme both to create chemically functional organogels and to reverse the process and break down these gels into their biologically compatible building blocks.”
In the experiments, researchers activated a sugar using a simple enzyme, which generated a compound that self-assembles into 3-D fibers measuring approximately 50 nanometers in diameter. As the fibers entangle, a large amount of solvent gets packed together, trapping some 10,000 molecules.
The resulting organogel materials could be used as biocompatible scaffolds for tissue engineering and designing membranes, according to Dordick. Other possible applications include delivery systems for pharmaceuticals and preservatives for food and cosmetics.
“The development of new materials that are molecularly defined and chemically functional at the nanoscale is of critical importance to biological applications such as drug delivery,” said Dordick. “We are finding the natural world has provided tools to create these materials without the need to generate new compounds that may be harmful to the body or environment.”
The findings are currently available online in advance of print publication July 17 by the journal Angewandte Chemie.
Dordick’s research involves using enzyme technology to produce unique chemical structures with applications in drug discovery, materials science, and chemical technology.
The research is led by Dordick and includes George John of the City University of New York; Guangyu Zhu, post-doctoral research associate at Rensselaer; and Jun Li of the University of Southern Mississippi. The paper is titled “Enzymatically Derived Sugar-Containing Self-Assembled Organogels with Nanostructured Morphologies.”
The funding for this research was provided by the National Science Foundation-funded Nanoscale Science and Engineering Center (NSEC) at Rensselaer, the Center for Directed Assembly of Nanostructures.
Nanotechnology at Rensselaer
In September 2001, the National Science Foundation selected Rensselaer as one of the six original sites for a new Nanoscale Science and Engineering Center (NSEC). As part of the U.S. National Nanotechnology Initiative, the program is housed within the Rensselaer Nanotechnology Center and forms a partnership between Rensselaer, the University of Illinois at Urbana-Champaign, and Los Alamos National Laboratory. The mission of Rensselaer’s Center for Directed Assembly of Nanostructures is to integrate research, education, and technology dissemination, and to serve as a national resource for fundamental knowledge in directed assembly of nanostructures. The five other original NSECs are located at Harvard University, Columbia University, Cornell University, Northwestern University, and Rice University.
Biotechnology and Interdisciplinary Studies at Rensselaer
At Rensselaer, faculty and students in diverse academic and research disciplines are collaborating at the intersection of the life sciences and engineering to encourage discovery and innovation. Rensselaer’s four biotechnology research constellations – biocatalysis and metabolic engineering, functional tissue engineering and regenerative medicine, biocomputation and bioinformatics, and integrative systems biology – engage a multidisciplinary mix of faculty and students focused on the application of engineering and physical and information sciences to the life sciences. Ranked among the world’s most advanced research facilities, the Center for Biotechnology and Interdisciplinary Studies at Rensselaer provides a state-of-the-art platform for collaborative research and world-class programs and symposia.
About Rensselaer
Rensselaer Polytechnic Institute, founded in 1824, is the nation’s oldest technological university. The university offers bachelor’s, master’s, and doctoral degrees in engineering, the sciences, information technology, architecture, management, and the humanities and social sciences. Institute programs serve undergraduates, graduate students, and working professionals around the world. Rensselaer faculty are known for pre-eminence in research conducted in a wide range of fields, with particular emphasis in biotechnology, nanotechnology, information technology, and the media arts and technology. The Institute is well known for its success in the transfer of technology from the laboratory to the marketplace so that new discoveries and inventions benefit human life, protect the environment, and strengthen economic development.
Media Contact
More Information:
http://ww.rpi.eduAll latest news from the category: Materials Sciences
Materials management deals with the research, development, manufacturing and processing of raw and industrial materials. Key aspects here are biological and medical issues, which play an increasingly important role in this field.
innovations-report offers in-depth articles related to the development and application of materials and the structure and properties of new materials.
Newest articles

A universal framework for spatial biology
SpatialData is a freely accessible tool to unify and integrate data from different omics technologies accounting for spatial information, which can provide holistic insights into health and disease. Biological processes…

How complex biological processes arise
A $20 million grant from the U.S. National Science Foundation (NSF) will support the establishment and operation of the National Synthesis Center for Emergence in the Molecular and Cellular Sciences (NCEMS) at…

Airborne single-photon lidar system achieves high-resolution 3D imaging
Compact, low-power system opens doors for photon-efficient drone and satellite-based environmental monitoring and mapping. Researchers have developed a compact and lightweight single-photon airborne lidar system that can acquire high-resolution 3D…





















