Simulated gene therapy

In a recent issue of The Journal of Chemical Physics, published by the American Institute of Physics (AIP), a group of researchers at the University of California, Berkeley and Los Alamos National Laboratory describe the first comprehensive, molecular-level numerical study of gene therapy.
Their work should help scientists design new experimental gene therapies and possibly solve some of the problems associated with this promising technique.
“There are several barriers to gene delivery,” says Nikolaos Voulgarakis of Berkeley, the lead author on the paper. “The genetic material must be protected during transit to a cell, it must pass into a cell, it must survive the cell's defense mechanisms, and it must enter into the cell's guarded nucleus.”
If all of these barriers can be overcome, gene therapy would be a valuable technique with profound clinical implications. It has the potential to correct a number of human diseases that result from specific genes in a person's DNA makeup not functioning properly — or at all. Gene therapy would provide a mechanism to replace these specific genes, swapping out the bad for the good. If doctors could safely do this, they could treat or even cure diseases like cystic fibrosis, certain types of cancer, sickle cell anemia, and a number of rare genetic disorders.
Safety is a primary concern when working with gene therapy. Some of the first attempts at gene therapy used viruses to insert DNA into cells — something that viruses naturally do anyway. Viruses can be dangerously toxic, however, and this fact was tragically demonstrated a decade ago when an 18-year-old boy enrolled in a gene therapy study had a massive immune reaction to the viruses used. He died just a few days into the treatment from multiple organ failure, precipitating an immediate halt to the trial.
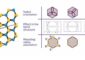
Since then, many alternatives to viruses have emerged for use in gene therapy, including synthetic molecules like “dendrimers,” a word that derives from the Greek word for “tree.” Similar to trees, dendrimers are branching molecules that are slightly positively charged. This allows them to be loaded with DNA (which is slightly negative charged) for insertion into a cell.
Dendrimers seem to offer many advantages over viruses. They may be much less toxic, and they may offer other advantages in terms of cost, ease of production, and the ability to transport very long genes. If they can be designed to efficiently — and safely — shuttle genes into human cells, then they may be a more practical solution to gene therapy than viruses.
So far, laboratory experiments with different types of dendrimers have shown that they can insert genes into cells, but only with very low efficiency. Hoping to discover the key to improving this efficiency, Voulgarakis and his colleagues simulated the detailed, atomic-level physical process of dendrimers entering cells. They varied parameters like the dendrimer size and the length of the DNA they carry. Modeling these parameters on a computer is a fast, inexpensive approach for testing different ideas and optimizing the delivery vehicle.
What they uncovered were the key factors that determine the success of dendrimers as gene delivery vehicles — things like the charges of the dendrimers and their target cell membranes, the length of DNA, and the concentration of surrounding salt. Their work has illuminated some of the molecular-level details that should help clinicians design the most appropriate gene vectors.
“Our study indicates that, over a broad range of biological conditions, the dendrimer/nucleic acid package will be stable enough to remain on the surface of the cell until translocation,” says Voulgarakis.
Dendrimers are also used clinically for delivering cancer drugs to tumors, and for helping to image the human body. In the future, Voulgarakis and his colleagues plan to study the possibility of using dendrimers as drug delivery vehicles.
The article ” Dendrimers as Synthetic Gene Vectors: Cell Membrane Attachment” by N. K. Voulgarakis, K. Ø. Rasmussen, and P. M. Welch was published in the April 21, 2009 issue of The Journal of Chemical Physics [J. Chem. Phys. 130, 155101 (2009)]. See: http://link.aip.org/link/?JCPSA6/130/155101/1 Journalists can obtain a free copy by emailing jbardi@aip.org.
ABOUT THE JOURNAL
The Journal of Chemical Physics, published by the American Institute of Physics (AIP), contains concise and definitive reports of significant research in methods and applications of chemical physics. Innovative research in traditional areas of chemical physics such as spectroscopy, kinetics, statistical mechanics, and quantum mechanics continue to be areas of interest to readers of JCP. In addition, newer areas such as polymers, materials, surfaces/interfaces, information theory, and systems of biological relevance are of increasing importance. See: http://jcp.aip.org.
ABOUT AIP
The American Institute of Physics (AIP) is a not-for-profit membership corporation chartered in 1931 for the purpose of advancement and diffusion of the knowledge of physics and its application to human welfare. An umbrella organization for 10 Member Societies, AIP represents over 134,000 scientists, engineers and educators and is one of the world's largest publishers of physics journals. A total-solution provider of publishing services, AIP also publishes 12 journals of its own (many of which have the highest impact factors in their category), two magazines, and the AIP Conference Proceedings series. Its online publishing platform Scitation (registered trademark) hosts more than 1,000,000 articles from more than 175 scholarly journals, as well as conference proceedings, and other publications of 25 learned society publishers.
Media Contact
More Information:
http://www.aip.orgAll latest news from the category: Physics and Astronomy
This area deals with the fundamental laws and building blocks of nature and how they interact, the properties and the behavior of matter, and research into space and time and their structures.
innovations-report provides in-depth reports and articles on subjects such as astrophysics, laser technologies, nuclear, quantum, particle and solid-state physics, nanotechnologies, planetary research and findings (Mars, Venus) and developments related to the Hubble Telescope.
Newest articles

A universal framework for spatial biology
SpatialData is a freely accessible tool to unify and integrate data from different omics technologies accounting for spatial information, which can provide holistic insights into health and disease. Biological processes…

How complex biological processes arise
A $20 million grant from the U.S. National Science Foundation (NSF) will support the establishment and operation of the National Synthesis Center for Emergence in the Molecular and Cellular Sciences (NCEMS) at…

Airborne single-photon lidar system achieves high-resolution 3D imaging
Compact, low-power system opens doors for photon-efficient drone and satellite-based environmental monitoring and mapping. Researchers have developed a compact and lightweight single-photon airborne lidar system that can acquire high-resolution 3D…