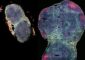
Scientists uncover new target in cancer mutation puzzle

In experiments with rodent and human cells, co-authors Mingxuan Xia, Ph.D., and Hartmut Land, Ph.D., explored how the Rho family of proteins, which are involved in cell movement, and thus in the progression from benign to malignant cancer, are controlled by two well-known cancer genes, p53 and Ras.
By closing in on this deadly collaboration, researchers showed for the first time why some molecules such as Rho are targeted by cancer genes – and how they might lead to a promising way to intervene against cancer.
“We have very little understanding of how Ras and p53 or any other potent gene mutations cooperate to cause malignant tumors,” said Land, who is professor and chair of the Department of Biomedical Genetics and scientific director of the James P. Wilmot Cancer Center at the University of Rochester Medical Center. “But we have suspected for a long time that the way to develop rational searches for new drug targets is to first understand how these oncogenes cooperate. And in this study we've shown for the first time that this idea might work.”
Land was among the scientists in the mid-1980s who first discovered oncogene collaboration. Since then, his work has focused on the complex interplay necessary for malignancy. And while other researchers, for example, are seeking ways to normalize a faulty gene such as p53, which is involved in half of all human tumors, Land's research group is taking the opposite approach. They are looking for ways to stop tumors by interfering with features of cancer cells dependent upon p53 and Ras mutations.
For cancer to develop, several mutations must arise and collaborate within a single cell. Ras and p53 mutations are particularly dangerous, implicated in colon, pancreas, lung and other cancers. Ras is part of a family that transmits signals controlling the way cells behave. Hyperactive Ras can lead to the uncontrolled growth of tumors. On the other hand, p53 is a tumor suppressor gene. Mutant p53 loses its ability to suppress cancer cells.
The UR team found that when cells were activated with Ras alone, the Rho proteins relocated to the cell membrane, their place of action, but remained inactive and did not cause cell movement. However, when active Ras was in cells that also had a p53 loss-of-function mutation, the Rho proteins became activated by Ras and promoted cell movement.
The experiments showed that when p53 is functioning properly it appears to be able to suppress the Ras signals to Rho, and thus shut down the movement of cancer cells. This was a previously unknown mechanism of action for the p53 gene.
Someday Rho may prove to be an attractive target for therapy because it is highly active only in malignant cells and not in normal cells.
“Now that we understand more about the role of Rho proteins as a target of cooperating cancer gene mutations in tumors with p53 mutations, we will look for other molecules with similar features,” Land said. “Our hope is that this line of research will give us a range of novel opportunities for treatments of cancer patients. We are at the beginning of a new and exciting road.”
Media Contact
More Information:
http://www.urmc.rochester.eduAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Red light therapy for repairing spinal cord injury passes milestone
Patients with spinal cord injury (SCI) could benefit from a future treatment to repair nerve connections using red and near-infrared light. The method, invented by scientists at the University of…

Insect research is revolutionized by technology
New technologies can revolutionise insect research and environmental monitoring. By using DNA, images, sounds and flight patterns analysed by AI, it’s possible to gain new insights into the world of…

X-ray satellite XMM-newton sees ‘space clover’ in a new light
Astronomers have discovered enormous circular radio features of unknown origin around some galaxies. Now, new observations of one dubbed the Cloverleaf suggest it was created by clashing groups of galaxies….