Strengthening thin-film bonds with ultrafast data collection
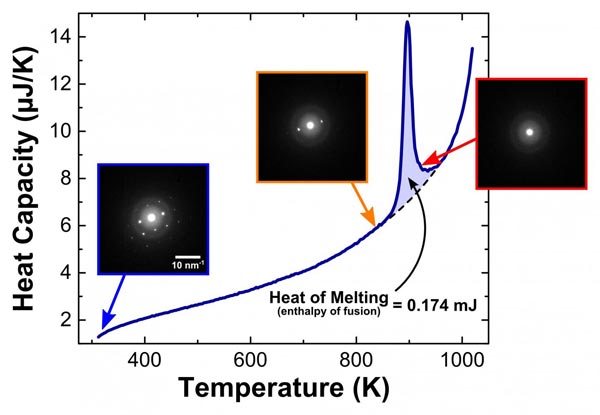
Temperature and structure: Graph shows heat absorbed by a thin film of aluminum as its temperature increased. Inset boxes show electron diffraction patterns captured by DTEM as temperature changes. The patterns reveal the crystal structure and orientation of the aluminum. At low temperatures, pattern is characteristic of a face-centered-cubic crystal structure. When the sample is heated past the large melting peak, the spots disappear indicating that the aluminum has lost its crystal structure due to melting. Credit: NIST
The combined device will help scientists study new materials and processes used to make advanced technologies, including state-of-the-art semiconductors and flat-screen display devices, says David LaVan, a NIST materials scientist who co-led the study.
Modern electronics manufacturing often pushes the limits of current measurement technology. Making a flat-screen display requires bonding a large sheet of a pure, rare material to an underlying metal substrate with as few defects as possible. To do so, manufacturers typically sandwich a thin film between the two materials and heat it rapidly to high temperatures, causing it to react and bond the metals.
This method usually works, but industry researchers would like to optimize the process. And existing tools to describe what's happening in the reactive thin film provide only incomplete information. One such technique, nanocalorimetry, can track very precisely large temperature changes—at rates up to ,1000 degrees Celsius per millisecond—that occur at a very small scale.
Such a measurement can alert researchers to a material's phase transitions, for example, when a metal melts. But nanocalorimetry tells researchers little about the actual chemical processes or microstructural changes they are measuring as a material heats up or cools down.
To study these changes, LaVan's LLNL collaborators Geoffrey Campbell, Thomas LaGrange and Bryan Reed developed a different device, the dynamic transmission electron microscope (DTEM). In traditional transmission electron microscopy, diffraction and transmission patterns made by electrons passing through a thin sample provide information about how the sample's atoms are arranged. But TEM typically requires that the sample maintain one crystal structure for an extended period, as the microscope's detector captures enough electrons to generate an image.
DTEM, by contrast, captures structural information very rapidly. It relies on a pulsed laser to send short, bright blasts of electrons through a sample. LaVan and his colleagues at NIST and Johns Hopkins realized that if the LLNL group's DTEM laser pulses were synched with a rapid temperature rise, the researchers could simultaneously track phase transitions and structural changes in materials they were studying. “It's like peanut butter and chocolate,” LaVan says. “If we can somehow get these two instruments working simultaneously, we'll have the whole story.”
But first the researchers needed to shrink the circuitry for their nanocalorimeter to a tenth of its original size, so that it could fit inside the microscope. The researchers also needed to write new software to synchronize the microscope's electron pulses with the nanocalorimeter's rapid heating pulses. “To get [the devices] to work together was really a substantial effort from three different research groups,” LaVan says.
Finally, LaVan and team member Michael Grapes, a research associate at NIST, and graduate student in materials science Timothy Weihs' group at Johns Hopkins, flew the redesigned nanocalorimeter to Livermore, synchronized it with the DTEM, and ran tests on thin films of materials such as aluminum, whose microstructural and thermal properties are well understood. The scientists found that, as expected, the nanocalorimeter recorded phase transitions at the same time the DTEM recorded structural changes, and both sets of measurements were consistent with their study materials' known properties.
The research team is already moving on to study other, less well-understood materials. Recently, the scientists have used their combined nanocalorimeter-DTEM to measure what happens when aluminum and nickel combine to form thin-film alloys. The team's study provides, for the first time, simultaneous structural and thermal data on this reaction at high heating rates, LaVan says.
* M.D. Grapes, T. LaGrange, L.H. Friedman, B.W. Reed, G.H. Campbell ,T.P. Weihs and D.A. LaVan. Combining nanocalorimetry and dynamic transmission electron microscopy for in situ characterization of materials processes under rapid heating and cooling. Review of Scientific Instruments 85, 084902. Published online Aug. 18, 2014.
Media Contact
All latest news from the category: Materials Sciences
Materials management deals with the research, development, manufacturing and processing of raw and industrial materials. Key aspects here are biological and medical issues, which play an increasingly important role in this field.
innovations-report offers in-depth articles related to the development and application of materials and the structure and properties of new materials.
Newest articles

A universal framework for spatial biology
SpatialData is a freely accessible tool to unify and integrate data from different omics technologies accounting for spatial information, which can provide holistic insights into health and disease. Biological processes…

How complex biological processes arise
A $20 million grant from the U.S. National Science Foundation (NSF) will support the establishment and operation of the National Synthesis Center for Emergence in the Molecular and Cellular Sciences (NCEMS) at…

Airborne single-photon lidar system achieves high-resolution 3D imaging
Compact, low-power system opens doors for photon-efficient drone and satellite-based environmental monitoring and mapping. Researchers have developed a compact and lightweight single-photon airborne lidar system that can acquire high-resolution 3D…





















