Cracked metal, heal thyself
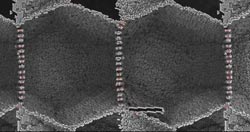
A computer simulation of the molecular stucture of a metal alloy, showing the boundaries between microcystalline grains (white lines forming hexagons), shows a small crack (dark horizontal bar just right of bottom center) that mends itself as the metal is put under stress. This simulation was one of several the MIT researchers used to uncover this new self-healing phenomenon. Simulation courtesy of Guoqiang Xu and Michael Demkowicz<br>
It was a result so unexpected that MIT researchers initially thought it must be a mistake: Under certain conditions, putting a cracked piece of metal under tension — that is, exerting a force that would be expected to pull it apart — has the reverse effect, causing the crack to close and its edges to fuse together.
The surprising finding could lead to self-healing materials that repair incipient damage before it has a chance to spread. The results were published in the journal Physical Review Letters in a paper by graduate student Guoqiang Xu and professor of materials science and engineering Michael Demkowicz.
“We had to go back and check,” Demkowicz says, when “instead of extending, [the crack] was closing up. First, we figured out that, indeed, nothing was wrong. The next question was: ‘Why is this happening?’”
The answer turned out to lie in how grain boundaries interact with cracks in the crystalline microstructure of a metal — in this case nickel, which is the basis for “superalloys” used in extreme environments, such as in deep-sea oil wells.
By creating a computer model of that microstructure and studying its response to various conditions, “We found that there is a mechanism that can, in principle, close cracks under any applied stress,” Demkowicz says.
Most metals are made of tiny crystalline grains whose sizes and orientations can affect strength and other characteristics. But under certain conditions, Demkowicz and Xu found, stress “causes the microstructure to change: It can make grain boundaries migrate. This grain boundary migration is the key to healing the crack,” Demkowicz says.
The very idea that crystal grain boundaries could migrate within a solid metal has been extensively studied within the last decade, Demkowicz says. Self-healing, however, occurs only across a certain kind of boundary, he explains — one that extends partway into a grain, but not all the way through it. This creates a type of defect is known as a “disclination.”
Disclinations were first noticed a century ago, but had been considered “just a curiosity,” Demkowicz says. When he and Xu found the crack-healing behavior, he says, “it took us a while to convince ourselves that what we’re seeing are actually disclinations.”
These defects have intense stress fields, which “can be so strong, they actually reverse what an applied load would do,” Demkowicz says: In other words, when the two sides of a cracked material are pulled apart, instead of cracking further, it can heal. “The stress from the disclinations is leading to this unexpected behavior,” he says.
Having discovered this mechanism, the researchers plan to study how to design metal alloys so cracks would close and heal under loads typical of particular applications. Techniques for controlling the microstructure of alloys already exist, Demkowicz says, so it’s just a matter of figuring out how to achieve a desired result.
“That’s a field we’re just opening up,” he says. “How do you design a microstructure to self-heal? This is very new.”
The technique might also apply to other kinds of failure mechanisms that affect metals, such as plastic flow instability — akin to stretching a piece of taffy until it breaks. Engineering metals’ microstructure to generate disclinations could slow the progression of this type of failure, Demkowicz says.
Such failures can be “life-limiting situations for a lot of materials,” Demkowicz says, including materials used in aircraft, oil wells, and other critical industrial applications. Metal fatigue, for example — which can result from an accumulation of nanoscale cracks over time — “is probably the most common failure mode” for structural metals in general, he says.
“If you can figure out how to prevent those nanocracks, or heal them once they form, or prevent them from propagating,” Demkowicz says, “this would be the kind of thing you would use to improve the lifetime or safety of a component.”
The work was funded by the BP-MIT Materials and Corrosion Center.
Written by: David L. Chandler, MIT News Office
Media Contact
More Information:
http://www.mit.eduAll latest news from the category: Materials Sciences
Materials management deals with the research, development, manufacturing and processing of raw and industrial materials. Key aspects here are biological and medical issues, which play an increasingly important role in this field.
innovations-report offers in-depth articles related to the development and application of materials and the structure and properties of new materials.
Newest articles

Making diamonds at ambient pressure
Scientists develop novel liquid metal alloy system to synthesize diamond under moderate conditions. Did you know that 99% of synthetic diamonds are currently produced using high-pressure and high-temperature (HPHT) methods?[2]…

Eruption of mega-magnetic star lights up nearby galaxy
Thanks to ESA satellites, an international team including UNIGE researchers has detected a giant eruption coming from a magnetar, an extremely magnetic neutron star. While ESA’s satellite INTEGRAL was observing…

Solving the riddle of the sphingolipids in coronary artery disease
Weill Cornell Medicine investigators have uncovered a way to unleash in blood vessels the protective effects of a type of fat-related molecule known as a sphingolipid, suggesting a promising new…





















