More evidence of cannabis-induced psychosis

Cannabis-based medicines given in a highly-controlled clinical environment unexpectedly lead to strong psychotic effects
Volunteers taking cannabis-based therapeutic drugs as part of a controlled trial, which had been approved by an ethics board as safe for the subjects, experienced psychotic effects just as strong as if they had smoked cannabis. These findings, highly unexpected in such a controlled environment, are published today in the peer-reviewed, Open Access journal BMC Psychiatry.
Dr Bernard Favrat and colleagues, from the Institut Universitaire de Medecine Legale in Switzerland, were conducting a clinical trial into the effects of orally administered delta-9-tetrahydrocannibol (THC), the active ingredient in cannabis, when two of their male subjects experienced impaired psychomotor functions and severe anxiety typical of cannabis-induced psychosis.
When smoking cannabis, the effects of THC on psychomotor functions usually start once the concentration in the blood has reached 10ng/ml plasma. The trial should have been safe as the subjects were given low doses of THC and had much lower concentrations in their blood. However the two male subjects experienced their reactions with blood concentrations of 4.7ng/ml and 6.2 ng/ml, respectively.
Favrat and colleagues found that both subjects reported severe anxiety and impaired psychomotor functions. Other effects included transient symptoms of derealisation and depersonalisation, and paranoid delusions. They were described by one subject as worse than those experienced after smoking cannabis. One subject was given dronabinol, a synthetic THC that has been in medical use in the USA since 1985. The other subject was asked to drink a decoction of natural THC. The authors hypothesise that the effect may have been because the THC had been ingested, rather than inhaled; digesting THC may produce potent THC metabolites, which induce psychotic effects.
Research into designing THC-based medications has boomed in the last few years, due to the many therapeutic effects of THC. These drugs could be used to alleviate muscle spasticity in multiple sclerosis patients, restore appetite in AIDS patients and alleviate pain and nausea in cancer patients undergoing chemotherapy. Such research has to be approved to ensure that it is treating patients ethically and safely. Dr Favrat’s research had been approved, which makes the findings even more unexpected.
Favrat and colleagues’ report adds to the body of evidence that cannabis might be more harmful than previously thought. In the UK, cannabis was downgraded to class C early last year, but government officials have called for a review of the decision following a series of studies revealing that cannabis dramatically increases the risk of developing mental illnesses.
Media Contact
More Information:
http://www.biomedcentral.comAll latest news from the category: Health and Medicine
This subject area encompasses research and studies in the field of human medicine.
Among the wide-ranging list of topics covered here are anesthesiology, anatomy, surgery, human genetics, hygiene and environmental medicine, internal medicine, neurology, pharmacology, physiology, urology and dental medicine.
Newest articles
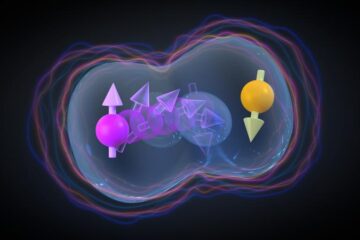
Experiment opens door for millions of qubits on one chip
Researchers from the University of Basel and the NCCR SPIN have achieved the first controllable interaction between two hole spin qubits in a conventional silicon transistor. The breakthrough opens up…
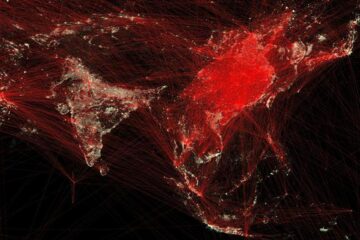
Economies take off with new airports
A global study by an SUTD researcher in collaboration with scientists from Japan explores the economic benefits of airport investment in emerging economies using nighttime satellite imagery. Be it for…
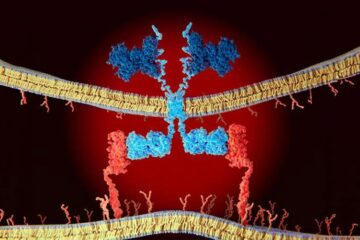
CAR T–cell immunotherapy targets
Pan-cancer analysis uncovers a new class of promising CAR T–cell immunotherapy targets. Scientists at St. Jude Children’s Research Hospital found 156 potential CAR targets across the brain and solid tumors,…





















