Spread of deadly eye cancer halted in cells and animals
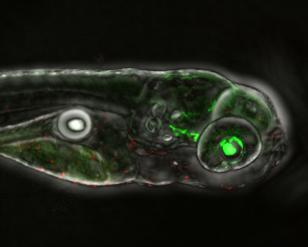
This is apoptosis induced by drug: Cell deaths (in red) induced by drug. Credit: Laura Asnaghi, Charles Eberhart
The new findings, the researcher say, offer a tempting target for treating the most common eye cancer in children — retinoblastoma — that originates in the retina. According to the World Health Organization, the cancer affects an estimated 7,000-8,000 children and kills up to 4,000 worldwide each year.
A report on the experiments was published Nov. 6th in the journal Oncogene.
“There is no effective treatment for retinoblastoma that spreads,” says Laura Asnaghi, Ph.D., M.Sc., a research associate faculty member in the Department of Pathology at the Johns Hopkins University School of Medicine.
“However, there is a chance for us to treat this deadly cancer if caught early before the tumors spread. Therefore, we looked into the causes for the tumor invasion, which can help us develop targeted therapies to prevent invasion.”
To uncover the series of molecular actions involved in tumor spread, the Johns Hopkins researchers started by analyzing tissues from 10 patients — five of the patients had invasive tumors and five had tumors that were not invasive. The researchers compared the RNA profiles of these two groups and found a twofold to threefold increase in RNA levels for the gene that codes for activin A receptor type 1C (ACVR1C) in invasive retinoblastoma cells compared to noninvasive cells.
This finding stood out because the activin receptor gene is already known to have a role in other cancers, including gallbladder and breast cancer. Researchers considered that the activin receptor may be a key target for suppressing cancer spread and growth in retinoblastoma.
Normally, when the activin receptor detects a growth signal, it triggers cells to grow and divide. The researchers treated cells with the drug SB505124, which blocks the activin receptor from detecting other growth signals, to see what would happen. They put the cells with the drug on a filter and measured invasion by looking at how many cells moved through the filter. Results showed that the growth, proliferation and invasion of retinoblastoma cells treated with the drug were suppressed by 60 to 80 percent.
After confirming the activin receptor's role in spreading retinoblastoma in cells, the researchers wanted to see whether this worked in live animals. They next pursued experiments in embryonic zebra fish, since this convenient model hasn't quite developed its immune systems yet and won't reject other types of cells transplanted into it.
The researchers injected human retinoblastoma cells into 2-day-old zebra fish eyes, and they monitored the growth and spread of the cancer cells by measuring the diameters of eye tumors over the next four to six days.
Then they administered the same drug (SB505124) used to inhibit the activin in the zebra fish eyes. According to the researchers, they saw a 55 percent reduction in the diameter of eye tumors compared to zebra fish eyes not injected with the drug. Overall, Asnaghi says, the experiments show that blocking the activin receptor could be effective in suppressing the growth and spread of invasive retinoblastoma cells in people.
“We hope our findings will provide new therapies for retinoblastoma, and lead to preserving vision and improving outcomes in a greater number of children affected by retinoblastoma both in the United States and worldwide,” says Asnaghi. “We are cautiously optimistic though, because we need to do more research before any related therapies can be safely developed or tested for patients.”
###
Other Johns Hopkins researchers involved in this study were David White, Nolan Key, Joshua Choi, Deepak Edward, Christopher Hurtado, Grace Lee, Jeff Mumm and
Charles Eberhart. Other researchers in the study include Alka Mahale, Hind Alkatan, Sahar Elkhamary, Saleh Al-Mesfer, Azza Maktab and Leen Abu Safieh of the King Khaled Eye Specialist Hospital, and Angel Carcaboso of Institut de Recerca Sant Joan de Déu.
The study is funded by the King Khaled Eye Specialist Hospital — Wilmer Eye Institute Collaborative Research Grant, the National Cancer Institute (Grant R21CA229919), the National Eye Institute (core grant EY001765) and The Jenny Fund.
COI: The researchers declare no conflicts of interest.
Media Contact
All latest news from the category: Health and Medicine
This subject area encompasses research and studies in the field of human medicine.
Among the wide-ranging list of topics covered here are anesthesiology, anatomy, surgery, human genetics, hygiene and environmental medicine, internal medicine, neurology, pharmacology, physiology, urology and dental medicine.
Newest articles

Microscopic basis of a new form of quantum magnetism
Not all magnets are the same. When we think of magnetism, we often think of magnets that stick to a refrigerator’s door. For these types of magnets, the electronic interactions…

An epigenome editing toolkit to dissect the mechanisms of gene regulation
A study from the Hackett group at EMBL Rome led to the development of a powerful epigenetic editing technology, which unlocks the ability to precisely program chromatin modifications. Understanding how…

NASA selects UF mission to better track the Earth’s water and ice
NASA has selected a team of University of Florida aerospace engineers to pursue a groundbreaking $12 million mission aimed at improving the way we track changes in Earth’s structures, such…





















