Protein involved in nematode stress response identified
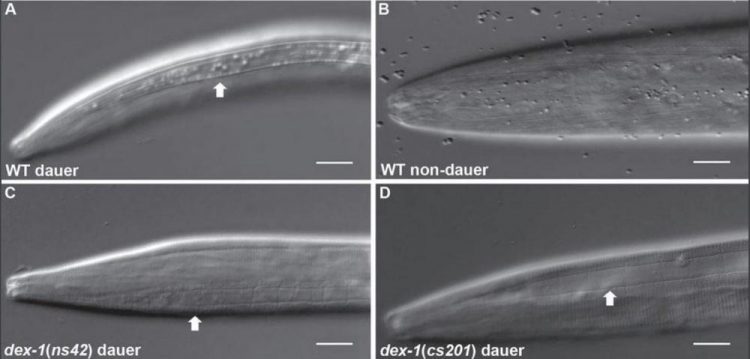
The free-living nematode, C. elegans, exists in a stress-resistant form called dauer in low-resource or overcrowded conditions. University of Illinois and University of Pennsylvania researchers found the protein responsible for this structural remodeling, DEX-1. The extracellular matrix protein may shed light on metastasis in human cancers and other processes involving stem cells. Credit: Nathan Schroeder, University of Illinois
Until now, it wasn't clear what molecular trigger was pulled to allow this structural remodeling in times of stress. But researchers at the University of Illinois and the University of Pennsylvania have discovered the protein responsible in the roundworm C. elegans.
“We're using a really simple animal system to understand basic biological questions that have implications not only for nematodes, including important crop parasites, but also for higher animals, including humans,” says Nathan Schroeder, assistant professor in the Department of Crop Sciences at U of I, and author of the new study published in Genetics.
When C. elegans larvae are stressed, they stop eating, their development halts, and they enter a stress-resistant stage known as dauer. In this form, their bodies become distinctly thinner and longer and develop an outer cuticle with ridges from tip to tail.
Schroeder and his team were investigating a protein called DEX-1 for an unrelated project when they noticed worms without the protein were “dumpy” in the dauer phase: they remained relatively short and round. Intrigued, the researchers decided to characterize the protein and its function in seam cells, the cells responsible for dauer remodeling.
“When we disrupted the DEX-1 protein, the seam cells did not remodel during dauer,” Schroeder says. “Seam cells have stem cell-like properties. We usually think about stem cells as controlling cell division, but we found that these cells are actually regulating their own shape through this protein, and that has an impact on overall body shape in response to stress.”
DEX-1 is an example of an extracellular matrix protein, a type that is extruded to form the mortar between cells. These proteins exist in every multicellular organism, not only keeping cells together but also facilitating interaction between cells. Not always in a good way; it turns out many extracellular matrix proteins, including a DEX-1 analogue, are associated with human diseases, such as metastatic breast cancer.
Schroeder says his group is interested in looking more closely at metastasis in cancers due to these proteins, but as a nematologist, he gets more excited about the prospect of understanding the basic biology and genetics of nematodes themselves, particularly parasitic species that affect crops.
“For many parasitic nematodes, when they're ready to enter the infective stage, they have a similar process. Many of the genes regulating the decision to go into or come out of that infective stage also regulate the decision to enter dauer,” he says. “This research gives us insight into their biology and how they make these developmental decisions.”
###
The article, “Epidermal remodeling in Caenorhabditis elegans dauers requires the nidogen domain protein DEX-1,” is published in Genetics [DOI: 10.1534/genetics.118.301557]. Authors include Kristen Flatt, Caroline Beshers, Cagla Unal, Jennifer Cohen, Meera Sundaram, and Nathan Schroeder. The research was supported by the National Institutes of Health.
Media Contact
More Information:
http://dx.doi.org/10.1534/genetics.118.301557All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Webb captures top of iconic horsehead nebula in unprecedented detail
NASA’s James Webb Space Telescope has captured the sharpest infrared images to date of a zoomed-in portion of one of the most distinctive objects in our skies, the Horsehead Nebula….
Cost-effective, high-capacity, and cyclable lithium-ion battery cathodes
Charge-recharge cycling of lithium-superrich iron oxide, a cost-effective and high-capacity cathode for new-generation lithium-ion batteries, can be greatly improved by doping with readily available mineral elements. The energy capacity and…
Novel genetic plant regeneration approach
…without the application of phytohormones. Researchers develop a novel plant regeneration approach by modulating the expression of genes that control plant cell differentiation. For ages now, plants have been the…





















