Genetic Regulation of the Thymus Function Identified
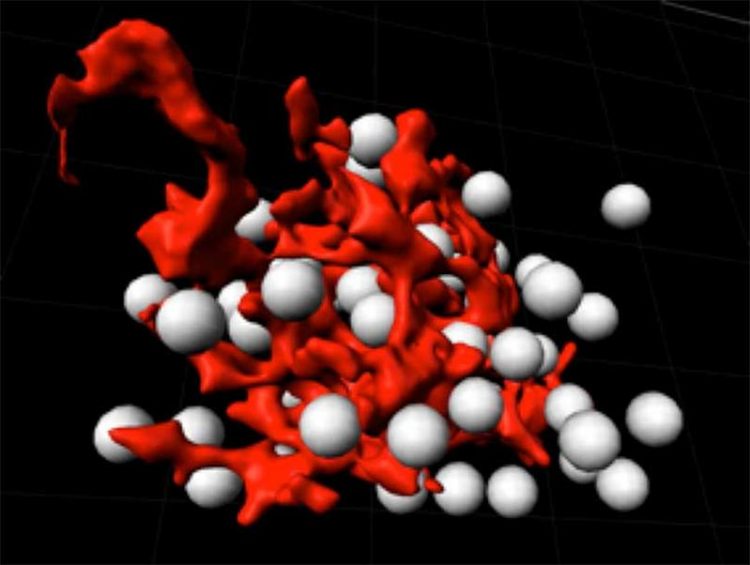
A single thymic epithelial cell (red) in contact with developing T cells (white). Department of Biomedicine, University of Basel
Like all vertebrate animals, humans use T cells in immune defense in order to protect the body against infections and malignant cells. Immune cells mature in the thymus, an organ located between the sternum and heart, during the embryonic stage, but they reach full functionality only when they interact with thymus epithelial cells – the teachers of the T cell, so to speak. The thymus begins to age from the second year of life.
More than 20 years ago, researchers discovered that the protein Foxn1 plays an important role in the development and differentiation of thymic epithelial cells. But it remained unclear which target genes controlled by Foxn1 were responsible for the formation and functions of the specialized epithelial cells. It was also unclear what Foxn1 actually did after development of the thymus in the embryo.
Healthy adults do not directly depend on the work of the thymus; however, the organ’s function becomes important again once the body requires new T cells – for example, after chemotherapy or a bone marrow transplant. To prevent the patient from remaining highly immune-deficient, thymic epithelial cells have to instruct the generation of new T cells.
Genome map of the regulator Foxn1
Researchers at the University of Basel, the University Children’s Hospital Basel and the University of Oxford have now succeeded in identifying in thymic epithelial cells the DNA sequence bound by the protein. The team, led by Professor Georg A. Holländer, was able to create a genome-wide map that lists all the DNA segments regulated by Foxn1. It emerged that Foxn1 controls almost all functions of the thymus, in whole or in part. “The protein is not only involved in the development of the organ, it also remains essential for its function throughout a person's life,” says the immunologist.
The research results provide important insights into the regulatory functions of thymic epithelial cells and could contribute to the development of new strategies for maintaining the thymus function in old age. “Now that we know exactly what Foxn1 does, we can think about how we can keep the thymus functioning in old age to reduce the risk of autoimmune disease and increased susceptibility to infections and tumors,” says Holländer.
Original article
Saulius Žuklys, Adam Handel, Saule Zhanybekova, Fatima Govani, Marcel Keller, Stefano Maio, Carlos E. Mayer, Hong Ying Teh, Katrin Hafen, Giuseppe Gallone, Thomas Barthlott, Chris P. Ponting and Georg A. Holländer
Foxn1 regulates key target genes essential for T cell development in postnatal thymic epithelial cells
Nature Immunology (2016), doi: 10.1038/ni.3537
Further information
Georg A. Holländer, University of Basel, Department of Biomedicine/University Children’s Hospital Basel, Tel. +41 61 270 50 69, email: georg-a.hollaender@unibas.ch
Saulius Zuklys, University of Basel, Department of Biomedicine/University Children’s Hospital Basel, Tel. +41 61 207 50 68, email: saulius.zuklys@unibas.ch
https://www.unibas.ch/en/News-Events/News/Uni-Research/Genetic-Regulation-of-the…
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Sea slugs inspire highly stretchable biomedical sensor
USC Viterbi School of Engineering researcher Hangbo Zhao presents findings on highly stretchable and customizable microneedles for application in fields including neuroscience, tissue engineering, and wearable bioelectronics. The revolution in…

Twisting and binding matter waves with photons in a cavity
Precisely measuring the energy states of individual atoms has been a historical challenge for physicists due to atomic recoil. When an atom interacts with a photon, the atom “recoils” in…

Nanotubes, nanoparticles, and antibodies detect tiny amounts of fentanyl
New sensor is six orders of magnitude more sensitive than the next best thing. A research team at Pitt led by Alexander Star, a chemistry professor in the Kenneth P. Dietrich…





















