Brushes in 3D
<br>
Polymer brushes are polymers in which individual polymer chains stand side by side on a surface, causing the chains to stick out like bristles on a brush.
In the journal Angewandte Chemie, American scientists have now presented a new simple method for making three-dimensional nanostructures in a controlled fashion from polymer brushes.
There are a wide variety of current and future applications for polymer brushes. For example, a coating of polymer brushes on a plastic surface such as an artificial heart valve or a dialysis machine can hinder the adsorption of proteins onto the surface. It can also be used in the fabrication of next-generation microelectronic devices. Other areas of application include biocompatible coatings for implants, chemical sensors, and new “intelligent” materials.
Although progress has been made with regard to new brush structures, current methods do not offer sufficient temporal and spatial control over the growth process. Usually, a self-organized monolayer of an initiator is assembled on a substrate and the polymer chains can grow out from there.
In order to obtain specific patterns, the initiator must be applied to the substrate in a corresponding pattern—a complex undertaking that is not manufacturable and does not allow for the generation of complex three-dimensional structures.
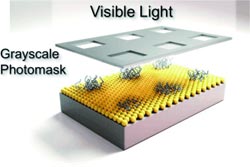
Craig J. Hawker and a team from the University of California, Santa Barbara, and The Dow Chemical Company (Midland, Michigan) have developed a new method that allows for the formation of brushes on a uniform initiator layer with both spatial and temporal control. Their simple method is based on a light-activated radical polymerization. The length of the bristles at any given location depends only on the duration and intensity of the local irradiation.
To form a specific structure, conventional photomasks can be used. These have openings in the areas to be irradiated and shield the other areas from the light. This allows for the formation of extensive patterns with submicrometer resolution in one step. All of this is made possible by a special iridium-based photocatalyst. It remains active for only a very short time after irradiation, so it cannot travel very far into nonirradiated areas while in its active state. It is even possible to use a grayscale photomask with continuously increasing opacity to produce gradated patterns.
Another advantage of this new method is that newly incorporated monomers are always added to the chain adjacent to the initiator, meaning that the initiator remains at the forward end of the growing chain. Because it is not destroyed as in other methods, and remains available at the right position, the polymerization can be stopped and restarted at any time. In this way the mask being used can be exchanged as often as desired. It is even possible to vary the monomer being used during the process. The complexity of accessible structures and applications is thus almost unlimited.
About the Author
Dr. Craig Hawker is the Alan and Ruth Heeger Professor of Interdisciplinary Studies at UCSB, and Director of the Dow Materials Institute and the California Nanosystems Institute. He is well known in the field of polymeric materials and has been honored with election as a Fellow of the Royal Society and the 2013 ACS Award in Polymer Chemistry.
Author: Craig J. Hawker, University of Califormia, Santa Barbara (USA), http://hawkergroup.mrl.ucsb.edu/craig-j-hawker
Title: Fabrication of Complex Three-Dimensional Polymer Brush Nanostructures through Light-Mediated Living Radical Polymerization
Angewandte Chemie International Edition, Permalink to the article: http://dx.doi.org/10.1002/anie.201301845
Media Contact
More Information:
http://pressroom.angewandte.orgAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Microscopic basis of a new form of quantum magnetism
Not all magnets are the same. When we think of magnetism, we often think of magnets that stick to a refrigerator’s door. For these types of magnets, the electronic interactions…

An epigenome editing toolkit to dissect the mechanisms of gene regulation
A study from the Hackett group at EMBL Rome led to the development of a powerful epigenetic editing technology, which unlocks the ability to precisely program chromatin modifications. Understanding how…

NASA selects UF mission to better track the Earth’s water and ice
NASA has selected a team of University of Florida aerospace engineers to pursue a groundbreaking $12 million mission aimed at improving the way we track changes in Earth’s structures, such…





















