NASA Simulation Portrays Ozone Intrusions From Aloft
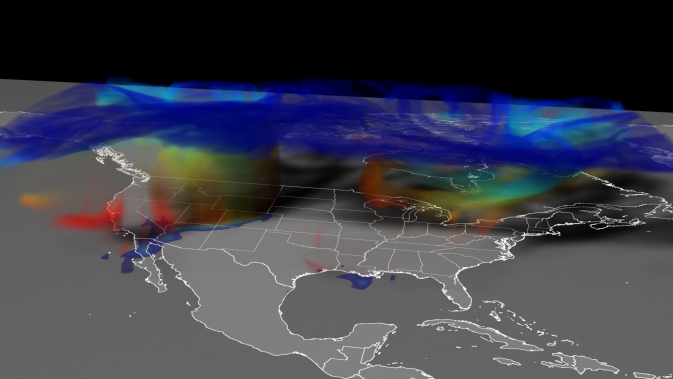
This GEOS-5 simulation of the April 6, 2012, stratospheric ozone intrusion shows ozone moving from high in the atmosphere, primarily in the stratosphere (blue), down close to the ground (red). On the ground, the highest concentrations of ozone (reds and oranges) track across Colorado. Image Credit: NASA Goddard's Scientific Visualization Studio
Ozone in the stratosphere, located on average 10 to 48 kilometers (6 to 30 miles) above the ground, typically stays in the stratosphere. Not on days like April 6, 2012.
On that day, a fast-moving area of low pressure moved northeast across states in the Western U.S., clipping western and northern Colorado. Ozone-rich stratospheric air descended, folding into tropospheric air near the ground.
Winds took hold of the air mass and pushed it in all directions, bringing stratospheric ozone to the ground in Colorado and along the Northern Front Range. The event, called a stratospheric ozone intrusion, raised ground-level ozone concentrations in some areas to potentially unhealthy levels. Watch the intrusion unfold in a new NASA simulation of the event.
Ozone high in the atmosphere, in the stratosphere, forms naturally when sunlight mingles with oxygen molecules to form the well-known “layer” that protects life on Earth from the sun's harmful ultraviolet rays. That's in contrast to ozone near the ground, in the troposphere, which forms from complex reactions involving chemicals emitted from industrial processes, vehicle exhaust, and other byproducts of fossil fuel combustion. Ozone at ground level can damage lung tissue and pose an immediate threat to sensitive groups such as people with asthma.
For this reason, the Clean Air Act requires the U.S. Environmental Protection Agency to set a threshold for ground-level ozone, as outlined in the National Ambient Air Quality Standards. States that exceed this threshold can be fined, although the EPA can grant exceptions for natural events or those proven to be beyond reasonable control.
That's why ozone intrusions are on the minds of air quality managers like Patrick Reddy, lead forecast meteorologist at Colorado's Department of Public Health and Environment in Denver, Colo. Reddy co-leads the EPA Stratospheric Intrusion Work Group, tasked to identify ozone intrusion events and collect input for improved analysis.
The state of Colorado flagged the concentrations associated with the April 6 event as possibly exceeding the EPA's allowable threshold. Now it's up to Reddy and colleagues to determine if the intrusion on April 6 is a viable candidate for the preparation of documentation to be classified as an exceptional event.
“We need to use the best science that we can to demonstrate conclusively that 'but for' this intrusion there would not have been an exceedence,” Reddy said.
Resolution Requirements
Reddy says it's fairly obvious when a stratospheric ozone intrusion has occurred, based on signatures in satellite data, air quality monitoring stations and meteorological data. For example, low water vapor, wind and high ozone at remote locations are often characteristic of stratospheric air.
Evidence of the intrusions, however, doesn't show up in the models currently used by air quality managers. Many of those models assume ozone moves from the stratosphere to the troposphere at a constant, average rate. This fails to capture the episodic intrusion events.
Meiyun Lin, an atmospheric scientist at Princeton University and NOAA's Geophysical Fluid Dynamics Laboratory (GFDL) in Princeton, New Jersey, set out to better quantify the impact of stratospheric ozone intrusions. Lin and colleagues used satellite and meteorological observations alongside a global chemistry-climate model to simulate intrusions in high-resolution.
Like the pixels in a photograph, the resolution of a model refers to the size of three-dimensional boxes of atmosphere. Models simulate the chemistry and atmospheric processes inside each box. For perspective, a model with 200-kilometer (124-mile) resolution is typical of today's high-end climate models, and 25-kilometer (16-mile) resolution is typical of high-end weather forecasts.
“We absolutely need to use a model with a grid size at least as small as, or smaller than, 50-by-50 kilometers (31-by-31 miles) to look at where and when the stratospheric air reaches the surface,” Lin said.
Lin's analysis, based on a GFDL model with 50-kilometer (31-mile) resolution, suggests that the impact on ground level ozone in the U.S. West from springtime intrusion events is two to three times greater than previously estimated. The study was published October 2012 in Journal of Geophysical Research.
Steven Pawson and Eric Nielsen, atmospheric scientists at NASA's Goddard Space Flight Center in Greenbelt, Md., are also in pursuit of improved model simulations of stratospheric ozone intrusions. The team set out to see if the Goddard Earth Observing System Model, Version 5 (GEOS-5) Chemistry-Climate Model could replicate stratospheric intrusions at 25-kilometer (16-mile) resolution.
They show that indeed, the model could replicate small-scale features, including finger-like filaments, within the apron of ozone-rich stratospheric air that descended over Colorado on April 6, 2012.
“High-resolution modeling is giving us the capability to examine these events comprehensively for the first time,” Nielsen said.
High-resolution models are possible due to computing power now capable of simulating the chemistry and movement of gasses and pollutants around the atmosphere and calculating their interactions. The addition of chemistry to these models, however, is not without a computational cost. For example, a weather forecast that takes about one hour of computational time would take five hours to run at the same resolution with the chemistry included.
“For a long time people thought excluding stratospheric chemistry was a reasonable approximation to make,” said Lesley Ott, an atmospheric scientist at NASA Goddard. “But recent work has shown that you really need to consider what the stratosphere is doing. It's not just something you can totally ignore, despite the fact that it's more computationally intensive.”
Atmospheric measurements from the ground and from aircraft suggest the higher resolution models are on track. In June and July 2011, NASA aircraft flew at low altitude over the Baltimore-Washington area as part of DISCOVER-AQ, a NASA airborne campaign to study urban air quality. Comparing data from the aircraft with the model output, Ott says the models performed well.
Tying it Together
Scientists already know that intrusions reaching surface air are more frequent in spring and early summer, when chemistry and weather conditions are more favorable for such events. Also, intrusions are more likely to affect mountainous regions in the U.S. West simply because land at elevation is closer to the stratosphere.
The next step is to find out how the frequency of intrusions changes from year to year and what controls its variability. “This is really the first time that our models are giving us the chance to try to answer those questions,” Ott said.
Reddy, too, looks forward to seeing if the models can streamline reporting and forecasting efforts. “The nice thing about the new model products is that they could help us potentially do a better job forecasting these events and documenting what happened for those events that we want to submit to the EPA,” he said.
The models could also help Reddy as his agency works to refine and expand its services. Models that could more accurately focus the timing and scale of intrusion effects would enhance the state's ability to issue advisories that better target affected populations.
Does that mean that spring skiers will have an additional forecast to consider before heading to the slopes?
“In the West, don't be surprised if on a clean-looking, windy day in spring there's an ozone health advisory,” Reddy said.
As for Bryan Duncan, an atmospheric scientist at NASA Goddard, “It wouldn’t stop me from enjoying the powder conditions.”
Related Links
› NASA's Global Modeling and Assimilation Office
› Colorado Department of Public Health and Environment
› NOAA's Geophysical Fluid Dynamics Laboratory
Kathryn Hansen
NASA's Earth Science News Team
Media Contact
All latest news from the category: Earth Sciences
Earth Sciences (also referred to as Geosciences), which deals with basic issues surrounding our planet, plays a vital role in the area of energy and raw materials supply.
Earth Sciences comprises subjects such as geology, geography, geological informatics, paleontology, mineralogy, petrography, crystallography, geophysics, geodesy, glaciology, cartography, photogrammetry, meteorology and seismology, early-warning systems, earthquake research and polar research.
Newest articles

Microscopic basis of a new form of quantum magnetism
Not all magnets are the same. When we think of magnetism, we often think of magnets that stick to a refrigerator’s door. For these types of magnets, the electronic interactions…

An epigenome editing toolkit to dissect the mechanisms of gene regulation
A study from the Hackett group at EMBL Rome led to the development of a powerful epigenetic editing technology, which unlocks the ability to precisely program chromatin modifications. Understanding how…

NASA selects UF mission to better track the Earth’s water and ice
NASA has selected a team of University of Florida aerospace engineers to pursue a groundbreaking $12 million mission aimed at improving the way we track changes in Earth’s structures, such…





















