Promising treatment for Huntington’s disease soon to be tested clinically
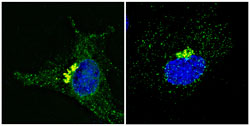
A protein that “releases” BDNF The nuclei in these two neuronal cells appear blue. The cell on the right has been treated with cysteamine, and so neuroprotective BDNF is progressively secreted from the Golgi apparatus, which appears yellow, and spreads around the nucleus within the cytoplasm. This reorganization results in better release of neuroprotective BDNF. © S. Humbert-F. Saudou/Institut Curie
At the Institut Curie, CNRS and Inserm researchers have shown that cysteamine, which is already used to treat a rare disease called cystinosis, prevents the death of neurons in Huntington’s disease. Like Alzheimer’s and Parkinson’s, Huntington’s disease, is characterized by the abnormal death of neurons.
Cysteamine raises neuronal levels of BDNF protein, a trophic factor which is depleted in Huntington’s disease, and by assaying BDNF in the blood it is possible to evaluate the effect of treatment. If other studies confirm these results, cysteamine could soon be used to treat Huntington’s disease, and BDNF could serve as a biomarker of its efficacy.
Huntington’s disease is a rare (1 in 10 000 people) neurological condition whose onset occurs between the ages of 35 and 50. The most striking symptoms are involuntary abnormal movements of the limbs, head and neck. These are accompanied by mental symptoms (anxiety, irritability, depression) and intellectual deterioration leading to dementia. Death occurs 15 to 20 years after disease onset as a result of complications (pulmonary embolism, pneumonia, other infection).
Clinical diagnosis is often difficult and time-consuming because the symptoms are highly variable and easy to confuse with psychological disorders. It must be confirmed by examination of the brain (by magnetic resonance imaging) or by genetic testing.
A mutant protein: huntingtin
Huntington’s disease is an autosomal dominant inherited disorder: if one of the two parents carries the mutant gene, 50% of the offspring will inherit this mutation and one day develop the disease. The IT15 gene responsible for the disease is located on chromosome 4 and encodes a protein called huntingtin, whose function is unknown. Normal huntingtin contains repeats of glutamine, an amino acid, but when there are more than 35 to 40 glutamines, huntingtin is considered to be mutant and induces the death of neurons, thereby causing the disease. Symptoms appear increasingly early as the number of glutamine repeats increases.
The same type of mutation causes other neurodegenerative diseases, each of which specifically affects different regions of the brain. In Huntington’s disease, there is progressive loss of neurons of the striatum, a region of the brain involved in the control of movement.
Thwarting apoptosis of neurons
At the Institut Curie, Frédéric Saudou and Sandrine Humbert(1) have already shown that brain-derived neurotrophic factor (BDNF)(2), when present in adequate amounts in the striatal neurons, blocks the effect of mutant huntingtin(3). Conversely, when BDNF levels decrease, the disease progresses. In patients with Huntington’s disease, BDNF levels in the striatal neurons are subnormal.
Under the direction of Frédéric Saudou and Sandrine Humbert, Maria Borell-Pagès has now shown in a mouse model of Huntington’s disease that cysteamine raises BDNF levels in striatal neurons. Cysteamine stimulates the secretion of BDNF, which explains its neuroprotective effect in different murine models of the disease. Cysteamine is already used clinically to treat a rare childhood disease called cystinosis(4).
The present study also demonstrates that BDNF, which can be assayed in blood, can be used as a biomarker. BDNF levels are decreased in animal models of Huntington’s disease, and are raised by cysteamine. Assay of BDNF in the blood should therefore enable evaluation of the efficacy of cysteamine treatment. A national, multicenter clinical trial is scheduled to begin by the end of 2006, and will test the effect of cysteamine and the value of BDNF as a biomarker in about 100 patients.
If these conclusions are confirmed clinically, cysteamine could become a routine treatment for Huntington’s disease.
Media Contact
All latest news from the category: Health and Medicine
This subject area encompasses research and studies in the field of human medicine.
Among the wide-ranging list of topics covered here are anesthesiology, anatomy, surgery, human genetics, hygiene and environmental medicine, internal medicine, neurology, pharmacology, physiology, urology and dental medicine.
Newest articles

‘Smart’ contact lenses could someday enable wireless glaucoma detection
Most people with early-stage glaucoma don’t know they have it, even though early treatment is key to reducing vision loss. While detecting a subtle increase in eye pressure helps doctors…

New tech may lead to smaller, more powerful wireless devices
Good vibrations… What if your earbuds could do everything your smartphone can do already, except better? What sounds a bit like science fiction may actually not be so far off….

Caution, hot surface!
An international research team from the University of Jena and the Helmholtz Institute Jena are demystifying the mechanisms by which high-intensity laser pulses produce plasma on the surface of solids….





















