Reversible Wound Closure
<br>
In first-aid situations, wounds must be quickly and effectively closed to stop blood loss and prevent infection. For treatment on arrival in a hospital, the temporary seal must be reopened, which often causes additional damage to the injured tissue. In the journal Angewandte Chemie, American scientists have now introduced a novel gel for sealing wounds. The gel can later be dissolved and gently removed.
Injuries sustained in remote areas, far from civilization, or in military action can often not be treated in a clinic until hours later. In such scenarios, a temporary wound closure system is desirable. Such a system should: 1) stop the bleeding for several hours, 2) adhere to the tissue, 3) be easy to apply, and 4) be easily removable in a controlled manner to make the wound accessible during surgical treatment. No single wound-closure systems currently available meet all of these requirements. Removal of blood-clotting agents or dressings requires tearing or surgical excision, both of which can increase the size of the wound and make it worse.
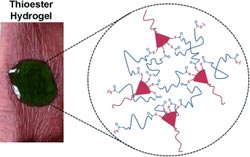
Scientists working at Boston University and the Beth Israel Deaconess Medical Center in Boston have now developed a wound-closure system based on a synthetic biocompatible gel that meets the requirements listed above. The gel is cross-linked through branched thioesters. The team, led by Mark W. Grinstaff, uses a chemical reaction known as thiol–thioester exchange in order to dissolve these gels for removal.
In this reaction, a thioester bond reacts with a thiolate anion to produce new thioester and thiolate products. The advantage of this reaction is that it takes place in an aqueous environment under physiological conditions. This type of reaction also occurs in natural biological processes. When the thioester gel is treated with cysteine methyl ester, the thioester bridges are rapidly split and the gel dissolves.
Wounds may be treated by simply mixing and applying two starting materials. The gel forms within seconds, adheres to the skin even when stress is applied and remains intact for several days. The gel absorbs any liquid exiting the wound. Treatment with cysteine methyl ester causes the wound closure to reopen within 30 minutes. To simulate injury to a vein, the researchers filled a section of bovine jugular vein with buffer solution and punctured it. Once the gel was applied, the damaged vein was completely sealed; after dissolution of the gel, the buffer solution flowed out again.
About the Author
Dr. Mark W. Grinstaff is Professor of Biomedical Engineering and Chemistry at Boston University and a College of Engineering Distinguished Faculty Fellow. His main interests are in the synthesis of new polymers and materials, and their application in medicine. He recently received the Edward M. Kennedy Award for Healthcare Innovation and was named a Fellow of the National Academy of Inventors.
Author: Mark W. Grinstaff, Boston University (USA), http://people.bu.edu/mgrin/
Title: A Dendritic Thioester Hydrogel Based on Thiol–Thioester Exchange as a Dissolvable Sealant System for Wound Closure
Angewandte Chemie International Edition, Permalink to the article: http://dx.doi.org/10.1002/anie.201308007
Media Contact
More Information:
http://pressroom.angewandte.orgAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Microscopic basis of a new form of quantum magnetism
Not all magnets are the same. When we think of magnetism, we often think of magnets that stick to a refrigerator’s door. For these types of magnets, the electronic interactions…

An epigenome editing toolkit to dissect the mechanisms of gene regulation
A study from the Hackett group at EMBL Rome led to the development of a powerful epigenetic editing technology, which unlocks the ability to precisely program chromatin modifications. Understanding how…

NASA selects UF mission to better track the Earth’s water and ice
NASA has selected a team of University of Florida aerospace engineers to pursue a groundbreaking $12 million mission aimed at improving the way we track changes in Earth’s structures, such…





















