‘Programmable’ Antibiotic Harnesses an Enzyme to Attack Drug-Resistant Microbes
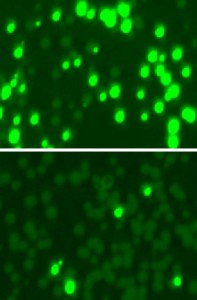
Subduing resistance: Researchers colonized mouse skin with a mix of bacterial cells, some resistant to the antibiotic kanamycin. They made the resistant cells glow (top) and treated the mix with an enzyme that targeted and killed off most resistant cells (bottom).
Researchers at Rockefeller University and their collaborators are working on smarter antibiotic. And in research published October 5 in Nature Biotechnology, the team describes a ‘programmable’ antibiotic technique that selectively targets the bad bugs, particularly those harboring antibiotic resistance genes, while leaving other, more innocent microbes alone.
“In experiments, we succeeded in instructing a bacterial enzyme, known as Cas9, to target a particular DNA sequence and cut it up,” says lead researcher Luciano Marraffini, head of the Laboratory of Bacteriology. “This selective approach leaves the healthy microbial community intact, and our experiments suggest that by doing so you can keep resistance in check and so prevent certain types of secondary infections, eliminating two serious hazards associated with treatment by classical antibiotics.”
The new approach could, for instance, reduce the risk of C. diff, a severe infection of the colon, caused by the Clostridium difficile bacterium, that is associated with prolonged courses of harsh antibiotics and is a growing public health concern.
The Cas9 enzyme is part of a defense system that bacteria use to protect themselves against viruses. The team co-opted this bacterial version of an immune system, known as a CRISPR (clustered regularly interspaced short palindromic repeats) system and turned it against some of the microbes.
CRISPR systems contain unique genetic sequences called spacers that correspond to sequences in viruses. CRISPR-associated enzymes, including Cas9, use these spacer sequences as guides to identify and destroy viral invaders.
The researchers directed Cas9 at targets of their choosing by engineering spacer sequences to match bacterial genes and then inserting these sequences into a cell along with the gene for Cas9. The cell’s own machinery then turns on the system. Depending on the location of the target in a bacterial cell, Cas9 may kill the cell or it may eradicate the target gene. In some cases, a treatment may prevent a cell from acquiring resistance, they found.
“We previously showed that if Cas9 is programmed with a target from a bacterial genome, it will kill the bacteria. Building on that work, we selected guide sequences that enabled us to selectively kill a particular strain of microbe from within a mixed population,” says first author David Bikard, a former Rockefeller postdoc who is now at the Pasteur Institute in Paris.
In initial experiments, Bikard and colleagues targeted a strain of the common skin and respiratory bacteria Staphylococcus aureus that is resistant to the antibiotic kanamycin. Treatment by Cas9 programmed to target a part of the resistance gene killed most of the resistant Staph, but left behind the kanamycin-susceptible Staph.
Targeted bacterial genocide is only one option. Bacteria share genes, including those conferring drug resistance, in the form of rings of DNA known as plasmids. In a second series of experiments, researchers turned Cas9 on tetracycline resistance-harboring plasmids in a strain of the potentially deadly multidrug resistant bacteria Staphylococcus aureus (MRSA).
Not only did the resistant cells become sensitive to tetracycline after Cas9 destroyed the plasmids, but the arrival of Cas9 in other Staph cells acted as an immunization, preventing them from taking on resistance-carrying plasmids.
And, in a final set of experiments, conducted in collaboration with Vincent Fischetti’s Laboratory of Bacterial Pathogenesis and Immunology, Chad Euler confirmed their test tube results on living skin, by using Cas9 to selectively kill kanamycin-resistant Staph infecting the shaved backs of mice.
In spite of the promising results, the delivery system needs improvement. The researchers used bacteria-infecting viruses to inject the programmed Cas9 enzymes into the bacterial cells, but these viruses only attack specific types of cells. Scientists need to devise a less discriminating method of delivery, before the technology can be used to develop a new class of antibiotics, Marraffini says.
In addition to its potential as a much-needed new weapon against drug-resistant microbes, the new system could also be used to advance research on the complex populations of microbes in the body, about which very little is known. “There are enormous microbial communities in the human body,” Marraffini says. “Programmable Cas9 enzymes may make it possible to analyze these populations by eliminating their members, one by one, and studying the effects.”
A National Institutes of Health Director’s New Innovator Award funded this research.
Contact Information
Zach Veilleux
212-327-8982
newswire@rockefeller.edu
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Microscopic basis of a new form of quantum magnetism
Not all magnets are the same. When we think of magnetism, we often think of magnets that stick to a refrigerator’s door. For these types of magnets, the electronic interactions…

An epigenome editing toolkit to dissect the mechanisms of gene regulation
A study from the Hackett group at EMBL Rome led to the development of a powerful epigenetic editing technology, which unlocks the ability to precisely program chromatin modifications. Understanding how…

NASA selects UF mission to better track the Earth’s water and ice
NASA has selected a team of University of Florida aerospace engineers to pursue a groundbreaking $12 million mission aimed at improving the way we track changes in Earth’s structures, such…





















