Oxygen in Place of Chlorine

Propylene oxide is an important bulk chemical that is used primarily in the production of polyurethane plastics.
Currently, propylene oxide is usually made from propylene (propene) in a process that uses chlorine as an oxidizing agent. This results in undesired byproducts as well as toxic chlorinated organic compounds. Existing alternative routes are mostly complicated and uneconomical.
The development of an environmentally friendly propylene oxide synthesis with oxygen as the oxidizing agent is high on the wish list. Japanese researchers have now developed a new catalyst that brings this goal closer. As the scientists working with Masatake Haruta report in the journal Angewandte Chemie, the catalyst is based on gold clusters and a special titanium-containing support.
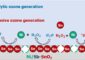
In the oxidation of propylene (propene, CH3–CH=CH2) to propylene oxide (propene oxide), an oxygen atom is formally inserted into the double bond. This forms a ring containing two carbon atoms and one oxygen atom. Using oxygen as the oxidizing agent had not been considered before because the oxygen molecule (O2) can only be split into individual oxygen atoms with the input of a large amount of energy. Furthermore, propylene preferentially reacts with atomic oxygen to form acrolein and not the desired propylene oxide. A suitable catalyst is eagerly sought, and has come to be viewed as the “holy grail” of catalyst research. There have been a number of catalytic developments that have been not quite satisfactory.
Building upon prior work, Haruta and his team have been able to achieve a further step. Their new catalyst consists of gold clusters, which are less than 2 nm in size, deposited on a special titanium-containing silicalite support. “It is important that the gold used is not in the form of nanoparticles, but is in clusters,” emphasizes Haruta. Although these two terms are often used interchangeably in the literature, there are important differences. Gold clusters are explicitly defined, structurally uniform nanoscopic structures, whereas gold nanoparticles are particles with size in the nanometer range that have neither uniform size nor structure. “Our gold clusters are able to convert oxygen and water into hydroperoxide species, which are transferred onto neighboring titanium centers,” explains Haruta. “The resulting titanium hydroperoxide species (Ti–OOH) are the actual reaction partners for the propylene, which is converted to propylene oxide.”
“The yields and selectivities we have achieved so far are inadequate for an industrial process,” says Haruta, “however, our catalyst is another important milestone on the way to an environmentally friendly synthesis for propylene oxide.”
Author: Masatake Haruta, Tokyo Metropolitan University (Japan), mailto:haruta-masatake@center.tmu.ac.jp
Title: Propene Epoxidation with Dioxygen Catalyzed by Gold Clusters
Angewandte Chemie International Edition 2009, 48, No. 42, 7862–7866, doi: 10.1002/anie.200903011
Media Contact
More Information:
http://pressroom.angewandte.orgAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Red light therapy for repairing spinal cord injury passes milestone
Patients with spinal cord injury (SCI) could benefit from a future treatment to repair nerve connections using red and near-infrared light. The method, invented by scientists at the University of…

Insect research is revolutionized by technology
New technologies can revolutionise insect research and environmental monitoring. By using DNA, images, sounds and flight patterns analysed by AI, it’s possible to gain new insights into the world of…

X-ray satellite XMM-newton sees ‘space clover’ in a new light
Astronomers have discovered enormous circular radio features of unknown origin around some galaxies. Now, new observations of one dubbed the Cloverleaf suggest it was created by clashing groups of galaxies….