Ancient mystery of cell division solved
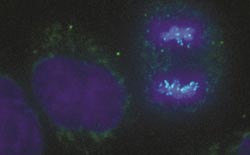
Dividing cell (right) with chromosomes colored in blue. Foto: Universität Göttingen <br>
An international team of scientists led by the University of Göttingen has deciphered a key mechanism of cell division. A striking feature of cell division is the condensation or compaction of chromosomes, which was first observed more than 130 years ago, but whose mechanism had been only vaguely understood so far. The Göttingen researchers successfully identified a force in a dividing cell that drives this process. The results were published in the journal Science.
The scientists at the Göttingen Centre for Molecular Biosciences led by Prof. Dr. Heinz Neumann used a trick from synthetic biology to equip cellular proteins with amino acids – the building blocks of proteins – that do not occur in nature. This endowed the proteins with particular properties, such as the ability to form cross-links with other proteins in the immediate vicinity upon irradiation.
Thereby, the researchers observed for the first time in an intact cell an interaction between nucleosomes, the basic building blocks of chromatin, and showed that this interaction plays a crucial role in the condensation of chromosomes during cell division. „Furthermore, we identified the mechanism of its regulation,“ explains Prof. Neumann: A signaling cascade triggers the interaction at the beginning of cell division and suppresses it again after its conclusion.
„Errors in the process could cause chromosomal aberrations, one of the main causes for the emergence of tumor cells,“ says Prof. Neumann. „With the discovery of the mechanisms and forces that control the condensation of chromosomes, new opportunities for the development of alternative therapeutic approaches to treat cancer may emerge.“
Original publication: Bryan J. Wilkins et al. A Cascade of Histone Modifications Induces Chromatin Condensation in Mitosis. Science 2014. Doi: 10.1126/science.1244508.
Contact:
Prof. Dr. Heinz Neumann
Georg-August University Göttingen
Faculty of Biology and Psychology
Free Floater Research Group „Applied Synthetic Biology“
Justus-von-Liebig-Weg 11, 37077 Göttingen, Germany
Phone +49 551 39-14088
Email: hneumann@uni-goettingen.de
Weitere Informationen:
http://www.uni-goettingen.de/en/121502.html
Free Floater Research Group „Applied Synthetic Biology“
Media Contact
More Information:
http://www.uni-goettingen.deAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Caution, hot surface!
An international research team from the University of Jena and the Helmholtz Institute Jena are demystifying the mechanisms by which high-intensity laser pulses produce plasma on the surface of solids….

Exploring the Asteroid Apophis With Small Satellites
In five years’ time, a large asteroid will fly very close to Earth – a unique opportunity to study it. Concepts for a national German small satellite mission are being…
First model of the brain’s information highways developed
Our human brain is not only bigger and contains more neurons than the brains of other species, but it is also connected in a special pattern: Thick bundles of neurons…





















