
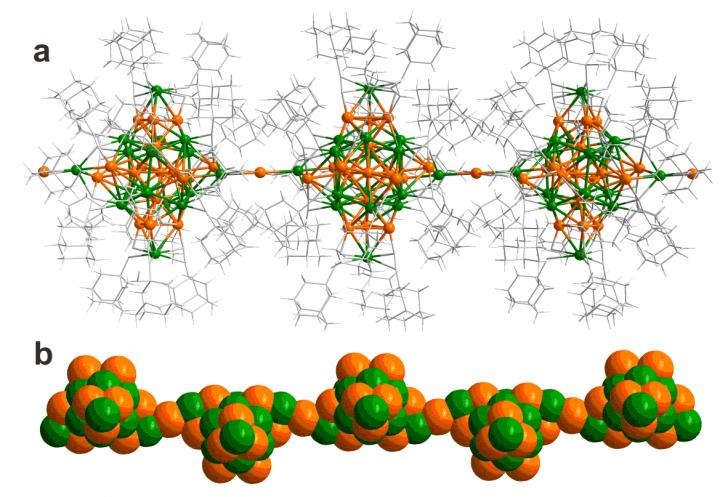
Figure a: Visualization of a linear polymer of the 34-atom silver-gold clusters with the inter-cluster metal-metal bonding in the horizontal direction (gold: orange, silver: green, ligand molecules (ethynyladamantane) are shown by grey sticks). Figure b: Shows the packing of metal atoms in the cluster polymer in a view rotated 90 degrees about the horizontal axis.
Credit: Peng Yuan/Xiamen University
The metal clusters were synthesized by means of wet chemistry adding gold and silver salts and ethynyladamantane molecules in a mixture of methanol and either chloroform or dichloromethane.
All syntheses produced the same 34-atom silver-gold clusters with an identical atomic structure, but surprisingly, the use of dichloromethane/methanol solvent initiated a polymerization reaction after cluster formation in solution and growth of human-hair-thick single crystals consisting of aligned polymeric chains of the clusters.
The crystals behaved as a semiconducting material in the direction of the polymer and as an electrical insulator in the cross directions. This behavior arises from metal-metal atomic bonding in the polymer direction while in the cross directions the metal clusters are isolated from each other by a layer of the ethynyladamantane.
Theoretical modeling of the cluster material by computer-intensive simulations using the density functional theory predicted that the material has an energy gap of 1.3 eV for electronic excitations.
This was confirmed by measurements of optical absorption and electrical conductivity in a layout where single crystals we mounted as part of a field-effect transistor, which showed a p-type semiconductor property of the material. Electrical conductivity along the polymer direction was about 1800-fold as compared to the cross directions.
“We were quite surprised by the observation that the polymer formation can be controlled by simple means of changing the solvent molecules. We discovered this probably by good luck, but we hope that this result can be applied in future to design hierarchical nanostructured materials with desired functionality”, says Professor Nanfeng Zheng from Xiamen University, who led the experimental work.
“This work shows an interesting example on how macroscopic material properties can be designed in the bottom-up synthesis of nanomaterials. Theoretical modeling of this material was quite challenging due to a large-scale model we had to build to account for the correct periodicity of the polymer crystal. To this end, we benefited very much of having access to some of the largest supercomputers in Europe”, says Academy Professor Hannu Hakkinen from the University of Jyvaskyla, who led the theoretical work.
###
The computer simulations were done in the Barcelona Supercomputing Center under a PRACE computing grant. The work by Hakkinen's group is supported by the Academy of Finland.
Link to the article in Nature Communications in 6th of May 2020: https:/
Article: P. Yuan et al.: “Solvent-mediated assembly of atom-precise gold-silver nanoclusters to semiconducting one-dimensional materials”, Nature Communications, published online May 6, 2020. DOI: 10.1038/s41467-020-16062-6
For more information:
Hannu Hakkinen, University of Jyvaskyla, Finland, hannu.j.hakkinen@jyu.fi, +358 400 247 973
Yang Cao, Xiamen University, China, yangcao@xmu.edu.cn Nanfeng Zheng, University of Xiamen, nfzheng@xmu.edu.cn
Communications officer Tanja Heikkinen, tanja.s.heikkinen@jyu.fi, tel. 358 50 581 8351
The Faculty of Mathematics and Science: https:/
Twitter: jyscience












