Surprising role of bacterial genes in evolution
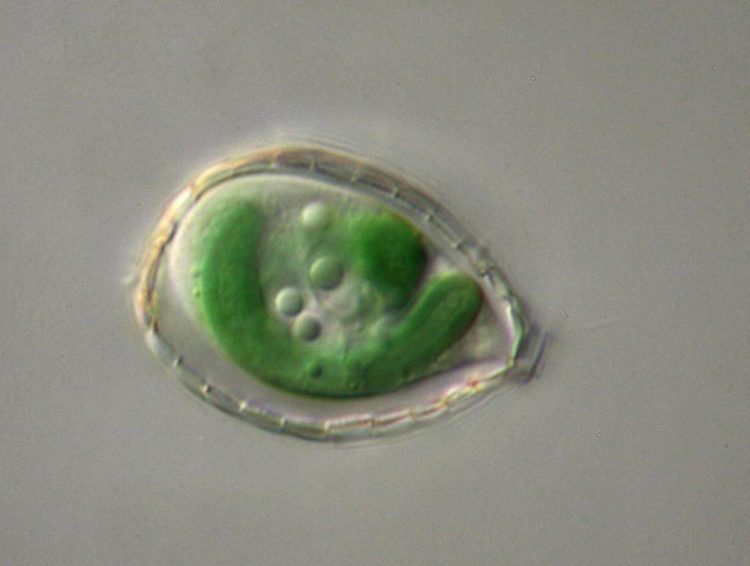
This micrograph of the single-celled Paulinella shows the the photosynthetic "machinery" in green. Credit: Eva Nowack
Sometimes a “host” organism can obtain genes from another organism that resides within its own cell (called an endosymbiont) through a process known as endosymbiotic gene transfer. At other times, an organism can obtain genes from a creature that lives in the surrounding environment, or from something that it eats, which is called horizontal gene transfer.
Furthermore, some levels of gene transfer can result in extensive loss of genes and genome reduction, especially for organisms that live as endosymbionts. For the first time, researchers have demonstrated that horizontal gene transfer may play a dominant role in compensating for genome reduction in an endosymbiont, and that this may be a key feature in the evolutionary transformation of an endosymbiont into an organelle (a longstanding organ inside a cell that often has specialized functions).
The research, published in the October 10 on-line edition of the Proceedings of the National Academy of Sciences, was conducted as a collaboration between scientists at Carnegie's Department of Plant Biology*, Rutgers University and Heinrich-Heine-Universität in Düsseldorf.
Specialized organelles called chloroplasts, which occur in plants and algae, are critical for performing the process of photosynthesis, and thus manufacturing sugars, starch and oils. These organelles originated more than 1 billion years ago when a photosynthetic bacterium, called a cyanobacterium, was engulfed by a host organism called a protist. There was a subsequent massive loss of genes from the genome of the cyanobacterium. Some of the lost cyanobacterial genes were transferred to the nucleus of the host cell through endosymbiotic gene transfer, while others were completely lost. This genome reduction was accompanied by the loss of many endosymbiont genes critical for the chain of enzymatic reactions involved in the biosynthesis of various essential compounds. How the host compensated for this gene reduction was only partially known.
Surprising results that shed light on the evolution of organelles from endosymbionts were obtained using the green, single-celled organism Paulinella chromatophora. Carnegie co-author Arthur Grossman explained: “We have recently proposed that the loss of genes from the photosynthetic organelle of P. chromatophora, which is called a chromatophore (originally an endosymbiotic cyanobacterium that was engulfed by a Paulinella species 60-200 million years ago), was in many cases compensated for by genes coming from neighboring bacteria in the environment. These new genes were integrated into the host nucleus and the proteins made from these genes were routed into chromatophores, where they compensate for the loss of genes.”
Lead author Eva Nowack remarked: “Of the at least 229 genes in the P. chromatophora nucleus that were acquired from various bacteria, only about 25% are of cyanobacterial origin and may have originated from endosymbiotic gene transfer. Excitingly, many of the remainder were acquired through horizontal gene transfer, representing genes from a variety of bacteria. Many of these bacteria-derived genes produce proteins that fill in gaps in chromatophore localized biosynthetic pathways. The original genes that filled the gaps were lost as a consequence of chromatophore genome reduction. This result suggests a dominant role for horizontal gene transfer in compensating for endosymbiont genome reduction.”
Furthermore, researchers found that a sister (ancestral) organism to P. chromatophora does not have a chromatophore and feeds on a variety of different bacteria, much like the way that white blood cells consume invading bacteria. In this new work, it is hypothesized that this method of feeding, called phagotrophy, may allow for the acquisition of different bacterial genes through horizontal gene transfer. In this way, the process of feeding facilitated bacterial gene selection as the cyanobacterial endosymbiont became a permanent resident within the phagotrophic host during early stages of chromatophore evolution.
###
* Authors on the paper are Eva C.M. Nowack, Dana C. Price, Debashish Bhattacharya, Anna Singer, Michael Melkonian, and Arthur R. Grossman This study was supported by National Science Foundation grant MCB-10370 (to A.R.G.), EF 08-27023 and OCE 11-29203 (to D.B.), and Deutsche Forschungsgemeinschaft Grant NO 1090/1-1 (to E.C.M.N.)
The Carnegie Institution for Science is a private, nonprofit organization headquartered in Washington, D.C., with six research departments throughout the U.S. Since its founding in 1902, the Carnegie Institution has been a pioneering force in basic scientific research. Carnegie scientists are leaders in plant biology, developmental biology, astronomy, materials science, global ecology, and Earth and planetary science.
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

High-energy-density aqueous battery based on halogen multi-electron transfer
Traditional non-aqueous lithium-ion batteries have a high energy density, but their safety is compromised due to the flammable organic electrolytes they utilize. Aqueous batteries use water as the solvent for…

First-ever combined heart pump and pig kidney transplant
…gives new hope to patient with terminal illness. Surgeons at NYU Langone Health performed the first-ever combined mechanical heart pump and gene-edited pig kidney transplant surgery in a 54-year-old woman…

Biophysics: Testing how well biomarkers work
LMU researchers have developed a method to determine how reliably target proteins can be labeled using super-resolution fluorescence microscopy. Modern microscopy techniques make it possible to examine the inner workings…





















