New way out: Researchers show how stem cells exit bloodstream
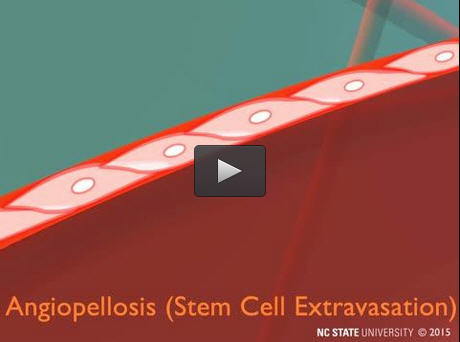
A stem cell exits the bloodstream through angiopellosis. Credit: Alice MacGregor Harvey, NC State University
When white blood cells need to get to the site of an infection, they can exit the bloodstream via a process called diapedesis. In diapedesis, the white blood cell changes its shape in order to squeeze between or through the epithelial cells that form the walls of the blood vessel.
Diapedesis is a well-understood process, and researchers believed that other types of cells, like therapeutic stem cells or even metastatic cancer cells, exited blood vessels in a similar way – with the cells pushing or squeezing themselves out.
But a group of researchers led by Ke Cheng, associate professor of molecular biomedical sciences at NC State with a joint appointment in the NC State/UNC-Chapel Hill Department of Biomedical Engineering, found that these stem cells behaved differently.
Therapeutic stem cells share the same ability to exit the bloodstream and target particular tissues that white blood cells do. But the precise way that they did so was not well understood, so Cheng and his team utilized a zebrafish model to study the process.
The genetically modified zebrafish embryos were transparent and had fluorescently marked green blood vessels. Researchers injected the embryos with white blood cells and cardiac stem cells from humans, rats and dogs. These cells had all been marked with a red fluorescent protein.
Through time-lapse three-dimensional light sheet microscopic imaging, Cheng and his team could trace the progress of these cells as they left the blood vessel. The white blood cells exited via diapedesis, as expected. When stem cells exited the blood vessel, however, the endothelial cells lining the vessel actively expelled them. Membranes surrounding the endothelial cells on either side of the stem cell stretched themselves around the stem cell, then met in the middle to push the stem cell out of the vessel.
“When you're talking about diapedesis, the white blood cell is active because it changes its shape in order to exit. The endothelial cells in the blood vessel are passive,” Cheng says. “But when we looked at therapeutic stem cells, we found the opposite was true – the stem cells were passive, and the endothelial cells not only changed their shape in order to surround the stem cell, they actually pushed the stem cells out of the blood vessel. We've named this process angiopellosis, and it represents an alternative way for cells to leave blood vessels.”
The researchers found two other key differences between angiopellosis and diapedesis: one, that angiopellosis takes hours, rather than minutes, to occur; and two, that angiopellosis allows more than one cell to exit at a time.
“Angiopellosis is really a group ticket for cells to get out of blood vessels,” Cheng says. “We observed clusters of cells passing through in this way. Obviously, this leads us to questions about whether other types of cells, like metastatic cancer cells, may be using this more effective way to exit the bloodstream, and what we may need to do to stop them.”
###
The research is published in Stem Cells. Tyler Allen, a graduate student in the comparative biomedical sciences program, is the first author of the paper. The research was supported by the National Institutes of Health and the American Heart Association.
-peake-
Note to editors: An abstract of the paper follows
“Angiopellosis as an alternative mechanism of cell extravasation”
DOI: 10.1002/stem.2451
Authors: Tyler Allen, Jhon Cores, Adam Vandergriff, Ke Cheng, NC State Department of Molecular Biomedical Sciences and joint Department of Biomedical Engineering, NC State and UNC-Chapel Hill; David Gracieux, Maliha Talib, Debra Tokarz, M. Taylor Hensley, James B.M. de Andrade, Phuong-Uyen Dinh, Jeffrey Yoder, North Carolina State University: Junnan Tang, First Affiliated Hospital of Zhengzhou University, Zhengzhou, Henan, China
Published: Online in Stem Cells
Abstract:
Stem cells possess the ability to home in and travel to damaged tissue when injected intravenously. For the cells to exert their therapeutic effect, they must cross the blood vessel wall and enter the surrounding tissues. The mechanism of extravasation injected stem cells employ for exit has yet to be characterized. Using intravital microscopy and a transgenic zebrafish line tg(Fli1a:egpf) with GFP-expressing vasculature, we documented the detailed extravasation processes in vivo for injected stem cells in comparison to white blood cells (WBCs). While WBCs left the blood vessels by the standard diapedesis process, injected cardiac and mesenchymal stem cells underwent a distinct method of extravasation that was markedly different from diapedesis. Here, the vascular wall undergoes an extensive remodeling to allow the cell to exit the lumen, while the injected cell remains distinctively passive in activity. We termed this process Angio-pello-sis, which represents an alternative mechanism of cell extravasation to the prevailing theory of diapedesis.
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Superradiant atoms could push the boundaries of how precisely time can be measured
Superradiant atoms can help us measure time more precisely than ever. In a new study, researchers from the University of Copenhagen present a new method for measuring the time interval,…

Ion thermoelectric conversion devices for near room temperature
The electrode sheet of the thermoelectric device consists of ionic hydrogel, which is sandwiched between the electrodes to form, and the Prussian blue on the electrode undergoes a redox reaction…

Zap Energy achieves 37-million-degree temperatures in a compact device
New publication reports record electron temperatures for a small-scale, sheared-flow-stabilized Z-pinch fusion device. In the nine decades since humans first produced fusion reactions, only a few fusion technologies have demonstrated…





















