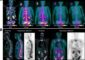
Self-plugging microneedles

Self-Plugging needles provided a more targeted, self-plugging drug delivery.
Credit: Terasaki Institute for Biomedical Innovation
… offer potential to improve delivery of drugs into the eyeball.
Published research on new eye treatment delivery shows improvement over hypodermic needles.
A collaborative team, including scientists from the Terasaki Institute for Biomedical Innovation (TIBI), has developed a novel, self-plugging microneedle for injecting therapeutics into the eyes, potentially solving one of the major challenges of treating eye diseases – accurate delivery of therapeutic drugs to the retina, while guarding against possible complications at the injection site.
Recently published in the journal Advanced Healthcare Materials, the research shows preclinical evidence that this innovative, biodegradable microneedle, dip-coated with a therapeutic drug for release upon insertion into the eyeball, can also be equipped with a special hydrogel that simultaneously seals off the insertion hole. Additionally, this self-plugging microneedle can be fabricated with different needle lengths, so therapeutics can be precisely targeted and dispersed to retinal tissues or other areas within the eyeball.
“This novel improvement in drug delivery treatment can avoid problems associated with using needles to treat serious eye diseases,” said Ali Khademhosseini, Ph.D., TIBI’s Director and CEO and a co-author of the study. “It is one of many simple, yet effective and impactful innovations from our institute, which is focused on translating biomedical engineering into clinical realities.”
The challenge – and need – for retinal drug delivery
The retina is a thin layer of cells that make up the inner lining of the very back of the eyeball. It enables vision by receiving light and converting it to signals that travel to the brain, which then interprets them as images.
As vital as it is to clear, crisp vision, the retina and its critical central portion, the macula, are also subject to different types of damage due to age, genetics, disease and other risk factors. This damage can cause symptoms ranging from distorted or seriously impaired vision to complete blindness and can be widespread among different populations. Indeed, age-related macular degeneration is the leading cause of irreversible vision loss in adults over 60, and diabetic retinopathy is the leading cause of blindness among U.S. working-aged adults aged 20–74 years.
Some forms of retinopathy can be treated by the injection of drugs into the eyeball using syringes and hypodermic needles. In this procedure, the injected drugs diffuse through the fluid within the eyeball to reach the damaged site. In many cases, multiple injections are needed over a prolonged period of time in order to protect their vision. In addition to this regimen being a hardship on patients, it introduces the risk of bacterial infection at the needle withdrawal site. It can also pose a risk for floating tumor cells in diseased eyes to escape through the puncture holes and migrate to other sites.
Previous approaches
In order to avoid these problems, implantable needles have been attempted for drug delivery; however, mechanical difficulties and navigation through the viscous fluid in the eyeball have hampered accurate needle placement and pose an additional risk of retinal damage. Other approaches involve polymer plugs to close the holes left by hypodermal needles, but this involves a separate step and time lag after injection.
Different types of microneedles, both in singular or patch form, have proven effective for drug delivery into different regions of the eye, with minimal puncture size and optimally targeted drug delivery. A collaborative team, including scientists from the Terasaki Institute for Biomedical Innovation, has built upon this technology by developing a novel, self-plugging microneedle for advanced injection treatment of the eyes.
The team utilized a precision fabrication method to create biodegradable, sharply-tipped, ultrathin microneedles of various lengths. This versatility in needle length allowed for a more controllable placement into the eyeball and more accurate drug delivery to the injury site; the fineness of the needles minimize incision size in the eyeball. The sharp end of the needle could be dip-coated with the therapeutic drug, with the needle releasing the drug upon insertion into the eyeball.
As an innovative addition, the blunt end of the microneedle was affixed with an experimentally-optimized hydrogel plug, which could swell after needle injection and seal off the insertion hole.
Preclinical testing
The two microneedle coatings were examined microscopically and tested for strength before subjecting them to validation tests for drug delivery and sealing effectiveness. Initial in vitro tests using the drug-loaded microneedles demonstrated almost complete drug delivery over a 24 hour period; similar experiments using dye-loaded SPNs inserted into vials of pig eyeball fluid demonstrated dye release over a suitable distance for injection into the eyeball.
Ex vivo experiments measuring the internal pressure of cultured pig eyeballs showed that there was almost no change in pressure after applications of SPNs, unlike the pressure drop obtained when non-plugging microneedles were applied. Examination of the tissue at the SPN application site confirmed firm attachment of the SPNs.
Further ex vivo experiments measured dye-tracked release from the SPNs into cultured rabbit and pig eyeballs. Tissue sections from these eyeballs revealed that dye distribution distance from the insertion site was directly proportional to needle length, demonstrating the spatially controllable drug delivery capabilities of the SPNs.
Final tests were conducted in vivo on pig animal models. Seven days after application of dye-loaded SPNs to the pigs’ eyes, observation through the pigs’ corneas revealed that the SPNs were still firmly implanted and there was no needle deformation. Leakage tests using surgical sponges showed no leakage of eyeball fluid.
Tissue section staining of the eyeballs exhibited complete dye dispersal and deep penetration into retinal tissues. Notably, there were no signs of inflammation or tissue disruption at the application site.
Authors are: KangJu Lee, SeungHyun Park, Dong Hyun Jo, Chang Sik Cho, Ha Young Jang, Jiyeon Yi, Minkyung Kang, Jaeho Kim, Ho Yun Jung, Jeong Hun Kim, WonHyoung Ryu, and Ali Khademhosseini.
This work was supported in part by a grant by the National Research Foundation of Korea (NRF), a grant funded by the Korean government (MSIT) (2021R1F1A105502211), and the Basic Science Research Program through the NRF, funded by the Ministry of Education (NRF-2017R1A6A3A04004741.
About The Terasaki Institute for Biomedical Innovation
The Terasaki Institute for Biomedical Innovation (terasaki.org) is a non-profit research organization that invents and fosters practical solutions that restore or enhance the health of individuals. Research at the Terasaki Institute leverages scientific advancements that enable an understanding of what makes each person unique, from the macroscale of human tissues down to the microscale of genes, to create technological solutions for some of the most pressing medical problems of our time. We use innovative technology platforms to study human disease on the level of individual patients by incorporating advanced computational and tissue-engineering methods. Findings yielded by these studies are translated by our research teams into tailored diagnostic and therapeutic approaches encompassing personalized materials, cells and implants with unique potential and broad applicability to a variety of diseases, disorders and injuries.
The Institute is made possible through an endowment from the late Dr. Paul I Terasaki, a pioneer in the field of organ transplant technology.
Contacts
For the Terasaki Institute
Judy Hopfield
jhopfield@terasaki.org
Media contact
Tim Ingersoll
tim@bioscribe.com
Media Contact
Stewart Han
Terasaki Institute for Biomedical Innovation
shan@terasaki.org
Office: 818-836-4393
All latest news from the category: Medical Engineering
The development of medical equipment, products and technical procedures is characterized by high research and development costs in a variety of fields related to the study of human medicine.
innovations-report provides informative and stimulating reports and articles on topics ranging from imaging processes, cell and tissue techniques, optical techniques, implants, orthopedic aids, clinical and medical office equipment, dialysis systems and x-ray/radiation monitoring devices to endoscopy, ultrasound, surgical techniques, and dental materials.
Newest articles

Properties of new materials for microchips
… can now be measured well. Reseachers of Delft University of Technology demonstrated measuring performance properties of ultrathin silicon membranes. Making ever smaller and more powerful chips requires new ultrathin…

Floating solar’s potential
… to support sustainable development by addressing climate, water, and energy goals holistically. A new study published this week in Nature Energy raises the potential for floating solar photovoltaics (FPV)…

Skyrmions move at record speeds
… a step towards the computing of the future. An international research team led by scientists from the CNRS1 has discovered that the magnetic nanobubbles2 known as skyrmions can be…