How does Parkinson's disease develop? Study raises doubts on previous theory of Parkinson’s disease
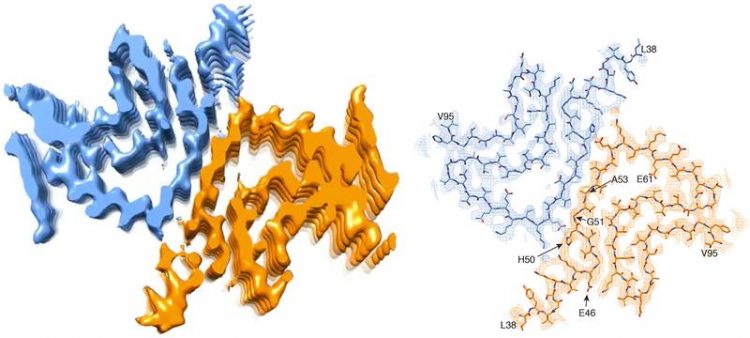
Cross section of an alpha-synuclein fibril. Left: 3D reconstruction of the fibril, showing two interacting protein molecules. Right: atomic model of the fibril structure. University of Basel
The arms and legs tremble incessantly, the muscles become weaker and the movements slower − these are typical symptoms that many Parkinson's patients suffer from. More than six million people are affected worldwide. In these patients, the dopamine-producing nerve cells in the brain slowly die off. The resulting lack of this neurotransmitter impairs motor function and often also affects the cognitive abilities.
Questionable: protein fibrils cause Parkinson's disease
So far, it was assumed that the protein alpha-synuclein is one of the trigger factors. This protein can clump together and form small needles, so-called fibrils, which accumulate and deposit as Lewy bodies in the nerve cells. These toxic fibrils damage the affected brain cells.
A team of scientists led by Prof. Henning Stahlberg from the Biozentrum of the University of Basel, in collaboration with researchers from Hoffmann-La Roche Ltd. and the ETH Zurich, have now artificially generated an alpha-synuclein fibril in the test tube. They have been able to visualize for the first time its three-dimensional structure with atomic resolution. “Contrary to our expectations, the results seem to raise more questions than they can hope to answer,” says Stahlberg.
It is important to know that in some congenital forms of Parkinson's disease, affected persons carry genetic defects in the alpha-synuclein gene. These mutations, it is suspected, eventually cause the protein to fold incorrectly, thus forming dangerous fibrils.
“However, our 3D structure reveals that a mutated alpha-synuclein protein should not be able to form these type of fibrils,” says Stahlberg. “Because of their location, most of these mutations would rather hinder the formation of the fibril structure that we have found.” In brief, if the fibril structure causes Parkinson's disease, the genetic defect would have to protect against the disease. But this is not the case. So, it could be possible that a different type of fibril or another form of the protein triggers the disease in these patients.
Study poses new questions
More investigations are now needed to understand this fibril structure. What are the effects of the alpha-synuclein mutations? Do they lead to distinct forms of protein aggregates? What is the role of the fibrils for the nerve cells, and why do these cells die? To date, the exact physiological function of alpha-synuclein is still not known. Since only the symptoms of this neurodegenerative disease can be alleviated with the current medications, new concepts are urgently needed.
Original source
Ricardo Guerrero-Ferreira, Nicholas M.I. Taylor, Daniel Mona, Philippe Ringler, Matthias E. Lauer, Roland Riek, Markus Britschgi, and Henning Stahlberg.
Cryo-EM structure of alpha-synuclein fibrils
eLife, published online 3 July 2018, doi: 10.7554/eLife.36402
Further information
Prof. Dr. Henning Stahlberg, University of Basel, Biozentrum, Tel. +41 61 387 32 62, E-Mail: henning.stahlberg@unibas.ch
https://elifesciences.org/articles/36402 Original source
Media Contact
More Information:
http://www.unibas.chAll latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

High-energy-density aqueous battery based on halogen multi-electron transfer
Traditional non-aqueous lithium-ion batteries have a high energy density, but their safety is compromised due to the flammable organic electrolytes they utilize. Aqueous batteries use water as the solvent for…

First-ever combined heart pump and pig kidney transplant
…gives new hope to patient with terminal illness. Surgeons at NYU Langone Health performed the first-ever combined mechanical heart pump and gene-edited pig kidney transplant surgery in a 54-year-old woman…

Biophysics: Testing how well biomarkers work
LMU researchers have developed a method to determine how reliably target proteins can be labeled using super-resolution fluorescence microscopy. Modern microscopy techniques make it possible to examine the inner workings…





















