Cohesin down-regulation drives hematopoietic stem cell aging
InflammAging affects hematopoietic stem cells (HSCs) through impairing their function and self-renewal by constant activation of cohesin-mediated inflammatory signals. [Graphic: Kerstin Wagner / FLI; Source: www.pixabay.de]
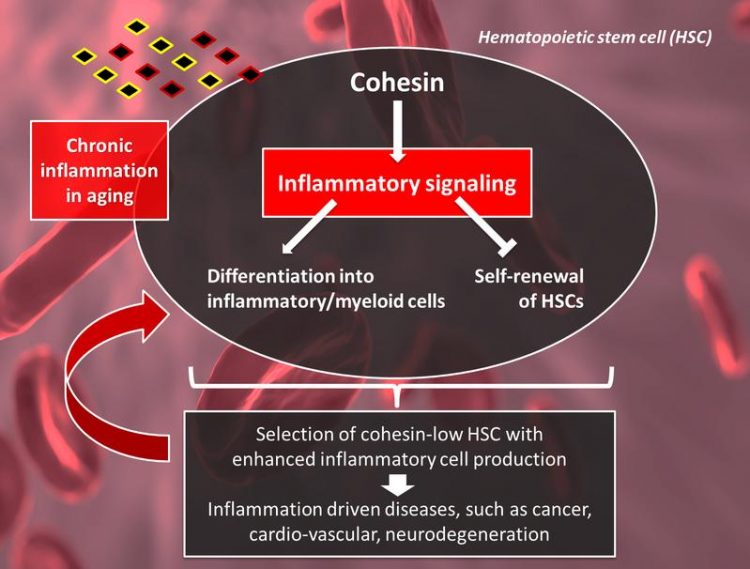
As the human body ages, inflammation increases and this accelerates the development of aging-associated diseases, a process called ‘InflammAging’. This process also affects the hematopoietic system in a way that inflammation instructs the stem cells of the system (hematopoietic stem cells, HSCs) to exhibit abnormal self-renewal and unbalanced differentiation into inflammatory/myeloid cells, which in turn leads to a further acceleration of InflammAging.
The drivers of stem cell aging are still incompletely understood. Gaining mechanistic insights into this process holds the promise to develop therapies to improve health during aging.
Researchers of the Leibniz Institute on Aging – Fritz Lipmann Institute (FLI) in Jena, Germany, have now found that the cohesin complex – a DNA clamp-holder – plays an important role in HSC response to inflammation and as such appears to drive key features of hematopoietic aging and InflammAging. The results are now published in the Journal of Experimental Medicine.
Hematopoietic system during aging and inflammation
A well working hematopoietic system is based on a balanced potential of the HSCs to differentiate into all important cell types of the blood system and to renew themselves as stem cells (self-renewal).
During aging and increasing, chronic inflammation, the balance of the hematopoietic system is lost. HCSs show impaired repopulation capacity and skewed differentiation: This means they are more likely to differentiate into inflammatory/myeloid cells than into lymphoid cells.
The transcription factor NF-κB controls many genes involved in inflammation. The pathway is activated in many inflammatory diseases. During aging, an increase in NF-κB signaling has been identified in HSCs. However, the functional consequences of enhanced NF-kB signaling on hematopoietic stem cells aging have not been worked out.
Cohesin as critical mediator of NF-κB signaling
Researchers of the Leibniz Institute on Aging (FLI) in Jena and their collaborators investigated aging and inflammation processes and their effects on the hematopoietic system.
“We knew that aging comes along with increased inflammation, but the consequences of chronic inflammation for hematopoietic stem cells and organism aging were incompletely understood”, Dr. Zhiyang Chen, postdoctoral researcher at FLI explains his motivation behind the study.
The current study increases our understanding and shows that cohesin mediates inflammatory signaling in HSCs and that this has functional consequences during aging and inflammatory processes.
Cohesin turns out to be important for normal differentiation of HSCs, but during aging and inflammation the same regulatory loop limits the stem cell function, especially the self-renewal capacity of the HSCs. Old HSCs fail to down-regulate cohesin-mediated inflammatory signals that remain chronically activated during aging.
“This mechanism of constant inflammation exhausts the stem cell pool and pushes the stem cells into faulty differentiation into inflammatory cells”, summarizes Prof. K. Lenhard Rudolph, senior group leader at FLI.
“In turn, these impairments increase the selection of abnormal stem cells with lower expression of cohesin that exhibit increases in self-renewal and mal-differentiation into inflammatory/myeloid cells – a hallmark feature of the hematopoietic system, which accelerates organism aging”.
Interfering with this molecular loop of chronic inflammation in aging hematopoietic stem cells could help to slow down aging and the development of aging-associated diseases such as cancer, cardiovascular and neurodegenerative diseases that are driven by increases in inflammation.
Publication
Cohesin-mediated NF-κB signaling limits hematopoietic stem cell self-renewal in aging and inflammation. Zhiyang Chen, Elias Moris Amro, Friedrich Becker, Martin Hölzer, Seyed Mohammad Mahdi Rasa, Sospeter Ngoci Njeru, Bing Han, Simone Di Sanzo, YuLin Chen, Duozhuang Tang, Si Tao, Ronny Hänold, Marco Groth, Vasily S. Romanov, Joanna M. Kirkpatrick, Johann M. Kraus, Hans A. Kestler, Manja Marz, Alessandro Ori, Francesco Neri, Yohei Morita, K. Lenhard Rudolph. J. Exp. Med. 2018, doi.org/10.1084/jem.20181505.
Contact
Dr. Kerstin Wagner
Leibniz Institute on Aging – Fritz Lipmann Institute (FLI), Beutenbergstr. 11, D-07745 Jena
Tel.: 03641-656378, Fax: 03641-656351, E-Mail: presse@leibniz-fli.de
Background information
The Leibniz Institute on Aging – Fritz Lipmann Institute (FLI) – upon its inauguration in 2004 – was the first German research organization dedicated to research on the process of aging. More than 330 employees from over 30 nations explore the molecular mechanisms underlying aging processes and age-associated diseases. For more information, please visit http://www.leibniz-fli.de.
The Leibniz Association connects 93 independent research institutions that range in focus from the natural, engineering and environmental sciences via economics, spatial and social sciences to the humanities. Leibniz Institutes address issues of social, economic and ecological relevance. They conduct knowledge-driven and applied basic research, maintain scientific infrastructure and provide research-based services. The Leibniz Association identifies focus areas for knowledge transfer to policy-makers, academia, business and the public. Leibniz Institutes collaborate intensively with universities – in the form of “WissenschaftsCampi” (thematic partnerships between university and non-university research institutes), for example – as well as with industry and other partners at home and abroad. They are subject to an independent evaluation procedure that is unparalleled in its transparency. Due to the institutes’ importance for the country as a whole, they are funded jointly by the Federation and the Länder, employing some 19,100 individuals, including 9,900 researchers. The entire budget of all the institutes is approximately 1.9 billion EUR. See http://www.leibniz-association.eu for more information.
Cohesin-mediated NF-κB signaling limits hematopoietic stem cell self-renewal in aging and inflammation. Zhiyang Chen, Elias Moris Amro, Friedrich Becker, Martin Hölzer, Seyed Mohammad Mahdi Rasa, Sospeter Ngoci Njeru, Bing Han, Simone Di Sanzo, YuLin Chen, Duozhuang Tang, Si Tao, Ronny Hänold, Marco Groth, Vasily S. Romanov, Joanna M. Kirkpatrick, Johann M. Kraus, Hans A. Kestler, Manja Marz, Alessandro Ori, Francesco Neri, Yohei Morita, K. Lenhard Rudolph. J. Exp. Med. 2018, doi.org/10.1084/jem.20181505.
http://www.leibniz-fli.de – Website Leibniz Institute on Aging – Fritz Lipmann Institute (FLI) Jena
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

High-energy-density aqueous battery based on halogen multi-electron transfer
Traditional non-aqueous lithium-ion batteries have a high energy density, but their safety is compromised due to the flammable organic electrolytes they utilize. Aqueous batteries use water as the solvent for…

First-ever combined heart pump and pig kidney transplant
…gives new hope to patient with terminal illness. Surgeons at NYU Langone Health performed the first-ever combined mechanical heart pump and gene-edited pig kidney transplant surgery in a 54-year-old woman…

Biophysics: Testing how well biomarkers work
LMU researchers have developed a method to determine how reliably target proteins can be labeled using super-resolution fluorescence microscopy. Modern microscopy techniques make it possible to examine the inner workings…





















