Liquid crystals in nanopores produce a surprisingly large negative pressure
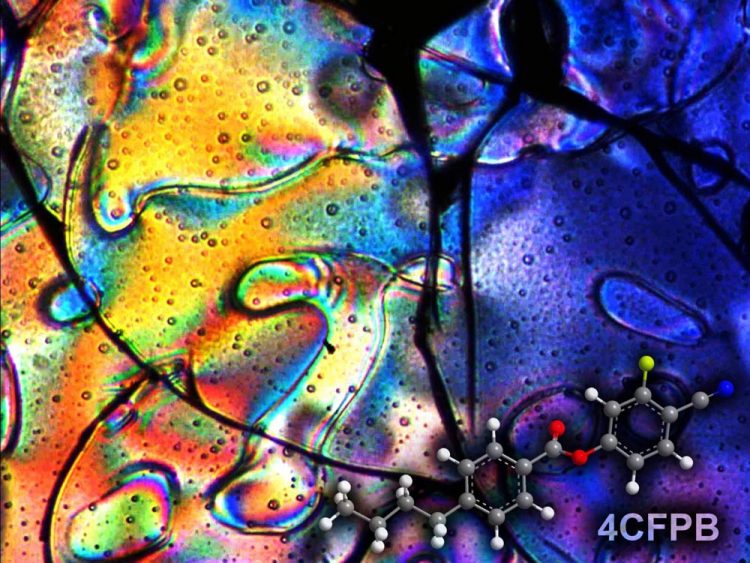
The negative pressure produced in nanopores by liquid crystals can significantly exceed 100 atmospheres. Above: The glass of the nematic phase of liquid crystal studied by scientists from the Institute of Nuclear Physics of the Polish Academy of Sciences in Cracow (Source: IFJ PAN). Source: IFJ PAN
At first glance, negative pressure appears to be an exotic phenomenon. In fact, it is common in nature, and what's more, occurs on many scales. On the scale of the Universe, the cosmological constant is responsible for accelerating the expansion of spacetime.
In the world of plants, attracting intermolecular forces (not: expanding thermal motions) guarantee the flow of water to the treetops of all trees taller than ten metres. On the quantum scale, the pressure of virtual particles of a false vacuum leads to the creation of an attractive force, appearing, for example, between two parallel metal plates (the famous Casimir effect).
“The fact that a negative pressure appears in liquid crystals confined in nanopores was already known. However, it was not known how to measure this pressure. Although we also cannot do this directly, we have proposed a method that allows this pressure to be reliably estimated,” says Dr. Tomasz Rozwadowski from the Institute of Nuclear Physics of the Polish Academy of Sciences (IFJ PAN) in Cracow, the first author of a publication in the Journal of Molecular Liquids.
The Polish physicists investigated a liquid crystal known as 4CFPB, made up of 1.67 nm long molecules with a molecular diameter of 0.46 nm. Experiments without nanopores, under normal and elevated pressure conditions (up to around 3000 atmospheres), were carried out at the University of Silesia in Katowice.
In turn, systems in silicon membranes with non-intersecting nanopores with a diameter of 6 and 8 nanometres were examined at the University of Leipzig (Germany). The geometry of the nanopores meant that there was room for only a few molecules of liquid crystal next to each other, with the long axes positioned along the walls of the channel.
The experiments looked at changes in various parameters of the liquid crystal (including dielectric dispersion and absorption). The measurements made it possible to conclude that an increase in pressure was accompanied by a slowing down of molecular mobility. However, the narrower the channels in which the molecules of liquid crystal in the nanopores were, the faster they moved.
The data also showed that the density of the liquid crystal molecules increased with increasing pressure whilst in the nanopores it decreased. There was also a change in the temperatures at which the liquid crystal passed from the liquid isotropic phase (with molecules arranged chaotically in space) to the simplest liquid crystalline phase (nematic; the molecules are still chaotically arranged, but they position their long axes in the same direction), and then to the glassy solid phase. As the pressure increased, the temperatures of the phase transitions increased. In the nanopores – they decreased.
“With increasing pressure, all the parameters of the liquid crystal we examined changed conversely to how they changed in nanopores with decreasing diameters. This suggests that the conditions in the nanopores correspond to a reduced pressure. Since the liquid crystal molecules in the channels try to stretch their walls, as if they were expanding, we can talk about negative pressure, relative to atmospheric pressure which constricts the walls,” says Dr. Rozwadowski.
The observed changes in physical parameters made it possible for the first time to estimate the value of the negative pressure appearing in the liquid crystal filling the nanopores. It turned out that (assuming the changes are linear) the negative pressure in nanopores can reach almost -200 atmospheres. This is an order of magnitude greater than the negative pressure responsible for water transport in trees.
“Our research is fundamental in nature, it provides information about the physics of phenomena occurring in liquid crystals constrained in nanopores of varying diameters. However, liquid crystals have many applications, for example in displays, optoelectronics, and medicine, so each new description of how these substances behave on the nanoscale in such specific spatial conditions may carry practical information,” stressed Dr. Rozwadowski.
###
Research on liquid crystals under spatial limitations was funded by the SONATA grant from the National Science Centre.
The Henryk Niewodniczanski Institute of Nuclear Physics (IFJ PAN) is currently the largest research institute of the Polish Academy of Sciences. The broad range of studies and activities of IFJ PAN includes basic and applied research, ranging from particle physics and astrophysics, through hadron physics, high-, medium-, and low-energy nuclear physics, condensed matter physics (including materials engineering), to various applications of methods of nuclear physics in interdisciplinary research, covering medical physics, dosimetry, radiation and environmental biology, environmental protection, and other related disciplines. The average yearly yield of the IFJ PAN encompasses more than 600 scientific papers in the Journal Citation Reports published by the Thomson Reuters. The part of the Institute is the Cyclotron Centre Bronowice (CCB) which is an infrastructure, unique in Central Europe, to serve as a clinical and research centre in the area of medical and nuclear physics. IFJ PAN is a member of the Marian Smoluchowski Kraków Research Consortium: “Matter-Energy-Future” which possesses the status of a Leading National Research Centre (KNOW) in physics for the years 2012-2017. The Institute is of A+ Category (leading level in Poland) in the field of sciences and engineering.
CONTACTS:
Dr. Tomasz Rozwadowski
The Institute of Nuclear Physics Polish Academy of Sciences
tel: +48 12 6628481
email: tomasz.rozwadowski@ifj.edu.pl
SCIENTIFIC PAPERS:
“Negative pressure effects on molecular dynamics and phase diagram of glass-forming nematic liquid crystal 4-cyano-3-fluorophenyl 4-butylbenzoate (4CFPB) confined in nanopores”
T. Rozwadowski, M. Massalska-Arodz, M. Jasiurkowska-Delaporte
Journal of Molecular Liquids 279 (2019) 127-132
DOI: https:/
LINKS:
http://www.
The website of the Institute of Nuclear Physics Polish Academy of Sciences.
http://press.
Press releases of the Institute of Nuclear Physics Polish Academy of Sciences.
IMAGES:
IFJ190424b_fot01s.jpg
HR: http://press.
The negative pressure produced in nanopores by liquid crystals can significantly exceed 100 atmospheres. Above: The glass of the nematic phase of liquid crystal studied by scientists from the Institute of Nuclear Physics of the Polish Academy of Sciences in Cracow (Source: IFJ PAN).
Media Contact
All latest news from the category: Physics and Astronomy
This area deals with the fundamental laws and building blocks of nature and how they interact, the properties and the behavior of matter, and research into space and time and their structures.
innovations-report provides in-depth reports and articles on subjects such as astrophysics, laser technologies, nuclear, quantum, particle and solid-state physics, nanotechnologies, planetary research and findings (Mars, Venus) and developments related to the Hubble Telescope.
Newest articles

Bringing bio-inspired robots to life
Nebraska researcher Eric Markvicka gets NSF CAREER Award to pursue manufacture of novel materials for soft robotics and stretchable electronics. Engineers are increasingly eager to develop robots that mimic the…

Bella moths use poison to attract mates
Scientists are closer to finding out how. Pyrrolizidine alkaloids are as bitter and toxic as they are hard to pronounce. They’re produced by several different types of plants and are…

AI tool creates ‘synthetic’ images of cells
…for enhanced microscopy analysis. Observing individual cells through microscopes can reveal a range of important cell biological phenomena that frequently play a role in human diseases, but the process of…





















