EXP2 protein helps deadliest malaria parasite obtain nutrients during infection
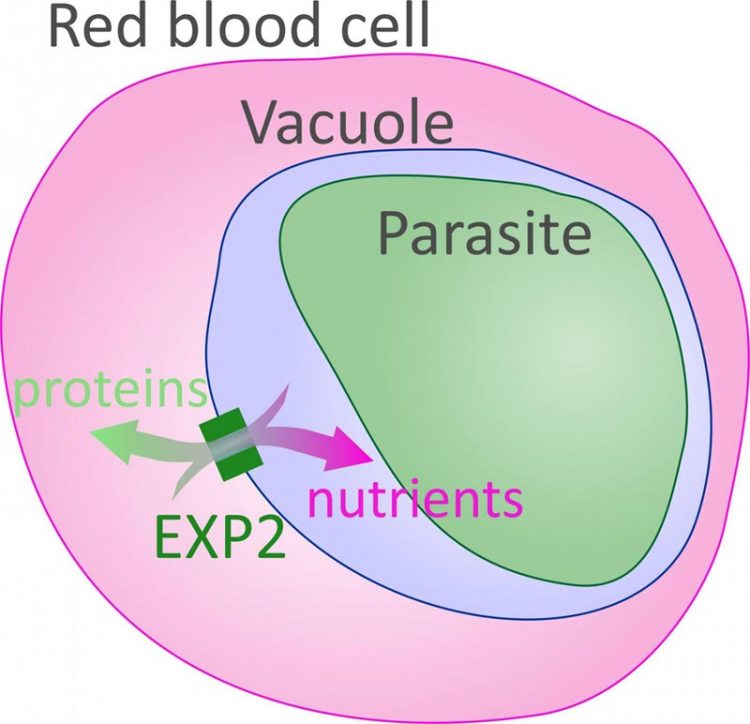
A group of proteins, called the Plasmodium translocon of exported proteins (PTEX), has been shown to be essential for transporting materials to and from the vacuole, the cellular compartment in which the malaria parasite resides. Researchers at the National Institutes of Health have determined that the PTEX protein EXP2 forms a channel in the vacuole membrane, which allows for passage of proteins and cellular nutrients to supply the parasite. Image courtesy of the Eunice Kennedy Shriver National Institute of Child Health and Human Development, National Institutes of Health
Researchers from the National Institutes of Health and other institutions have deciphered the role of a key protein that the malaria parasite Plasmodium falciparum uses to obtain nutrients while infecting red blood cells. Their study appears in Nature Microbiology.
According to the World Health Organization, in 2016 there were an estimated 216 million malaria cases and 445,000 malaria deaths. P. falciparum is responsible for most malaria-related deaths globally.
The parasite remodels the red blood cell it infects to obtain nutrients. During this process, the parasite secretes hundreds of proteins that need to be transported from the vacuole, the compartment in which the parasite resides, to the interior of the cell.
A group of proteins, called the Plasmodium translocon of exported proteins (PTEX), has been shown to be essential for transporting materials to and from the vacuole.
Previous studies have uncovered the function of one of the proteins in the PTEX group to reshape proteins for transport, but the function of other proteins in the group have not been well understood.
In the current study, researchers analyzing blood cell cultures from healthy people determined that the PTEX protein EXP2 forms a channel in the vacuole membrane, which allows for passage of proteins and cellular nutrients to supply the parasite.
The researchers hope that their discovery will lead to the development of new drugs to prevent formation of the channel and block the transport of nutrients and proteins to the parasite.
###
WHO:
Joshua Zimmerberg, M.D., Ph.D., chief of the Section on Integrative Biophysics at NIH's Eunice Kennedy Shriver National Institute of Child Health and Human Development, is available for comment.
ARTICLE:
Garten, M. EXP2 is a nutrient-permeable channel in the vacuolar membrane of Plasmodium and is essential for protein export via PTEX. Nature Microbiology. 10.1038/s41564-018-0222-7
About the Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD): NICHD conducts and supports research in the United States and throughout the world on fetal, infant and child development; maternal, child and family health; reproductive biology and population issues; and medical rehabilitation. For more information, visit NICHD's website.
About the National Institutes of Health (NIH): NIH, the nation's medical research agency, includes 27 Institutes and Centers and is a component of the U.S. Department of Health and Human Services. NIH is the primary federal agency conducting and supporting basic, clinical, and translational medical research, and is investigating the causes, treatments, and cures for both common and rare diseases. For more information about NIH and its programs, visit http://www.
Media Contact
All latest news from the category: Health and Medicine
This subject area encompasses research and studies in the field of human medicine.
Among the wide-ranging list of topics covered here are anesthesiology, anatomy, surgery, human genetics, hygiene and environmental medicine, internal medicine, neurology, pharmacology, physiology, urology and dental medicine.
Newest articles

Bringing bio-inspired robots to life
Nebraska researcher Eric Markvicka gets NSF CAREER Award to pursue manufacture of novel materials for soft robotics and stretchable electronics. Engineers are increasingly eager to develop robots that mimic the…

Bella moths use poison to attract mates
Scientists are closer to finding out how. Pyrrolizidine alkaloids are as bitter and toxic as they are hard to pronounce. They’re produced by several different types of plants and are…

AI tool creates ‘synthetic’ images of cells
…for enhanced microscopy analysis. Observing individual cells through microscopes can reveal a range of important cell biological phenomena that frequently play a role in human diseases, but the process of…





















