Alzheimer’s disease; new approach, new possibilities?

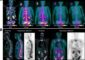
Scientists from the Flanders Interuniversity Institute for Biotechnology (VIB) associated with the University of Antwerp have achieved a new breakthrough in their research on the origins of Alzheimer’s disease. Their alternative approach opens up new prospects for developing a treatment which can slow the disease’s progress. The researchers have shown that ´the plaques´ which form in the brain of patients are linked to damage to nearby blood vessels. Leakage appears to occur between the blood vessels and the brain, as a result of which the plaques develop and the disease manifests itself. This research is published today in the ‘American Journal of Pathology’.
Alzheimer’s disease
Alzheimer’s disease, a degenerative disease that gradually and progressively destroys brain cells, affects between 50% and 70% of all dementia patients and is therefore the major form of dementia. About 100,000 people suffer from this disease in Belgium. The damage caused to memory and mental functioning makes it one of today’s most frightening syndromes. In particular, the first realization of the loss of any sense of reality is extremely difficult to accept. So, science continues to search feverishly for ways to treat the disease.
The formation of plaques plays a key role
Alzheimer’s disease is characterized by an increasing deposit of the amyloid-ß protein in the brain. The accumulation of this protein results in ´plaques´; deposits which settle in the brain cells responsible for memory and observation. How the plaques develop is the key in the search for a treatment. Samir Kumar-Singh and his colleagues on a team headed by Christine Van Broeckhoven have unraveled how certain plaques are formed. In various mouse models, they demonstrate that the plaques attach primarily onto the blood vessels. The vessels show clear structural damage, so that the strictly-controlled separation between blood vessels and brain is compromised and leakage occurs.
A new model as a first step towards a treatment?
Under normal circumstances, the blood vessels transport the excess amyloid-ß protein away from the brain. However, the protein has a harmful effect on blood vessel walls. This effect is perhaps strengthened as a result of ageing, which causes the protein to be removed less efficiently. The blood vessel loses strength and in its immediate vicinity the accumulation of the amyloid-ß protein increases and plaques develop. Finally, the damage to the blood vessel is so great that it is no longer functional and other blood vessels take over its tasks.
The results of the research of Samir Kumar-Singh opens up alternatives for developing new treatments. For example, a treatment which promotes the removal of the amyloid-ß protein from the brain can significantly impede the onset of Alzheimer’s disease. A new approach which might have far-reaching consequences. Additional research should make it possible to verify this in greater detail.
Media Contact
More Information:
http://www.vib.beAll latest news from the category: Medical Engineering
The development of medical equipment, products and technical procedures is characterized by high research and development costs in a variety of fields related to the study of human medicine.
innovations-report provides informative and stimulating reports and articles on topics ranging from imaging processes, cell and tissue techniques, optical techniques, implants, orthopedic aids, clinical and medical office equipment, dialysis systems and x-ray/radiation monitoring devices to endoscopy, ultrasound, surgical techniques, and dental materials.
Newest articles

Properties of new materials for microchips
… can now be measured well. Reseachers of Delft University of Technology demonstrated measuring performance properties of ultrathin silicon membranes. Making ever smaller and more powerful chips requires new ultrathin…

Floating solar’s potential
… to support sustainable development by addressing climate, water, and energy goals holistically. A new study published this week in Nature Energy raises the potential for floating solar photovoltaics (FPV)…

Skyrmions move at record speeds
… a step towards the computing of the future. An international research team led by scientists from the CNRS1 has discovered that the magnetic nanobubbles2 known as skyrmions can be…