Urinary tract infection: How bacteria nestle in
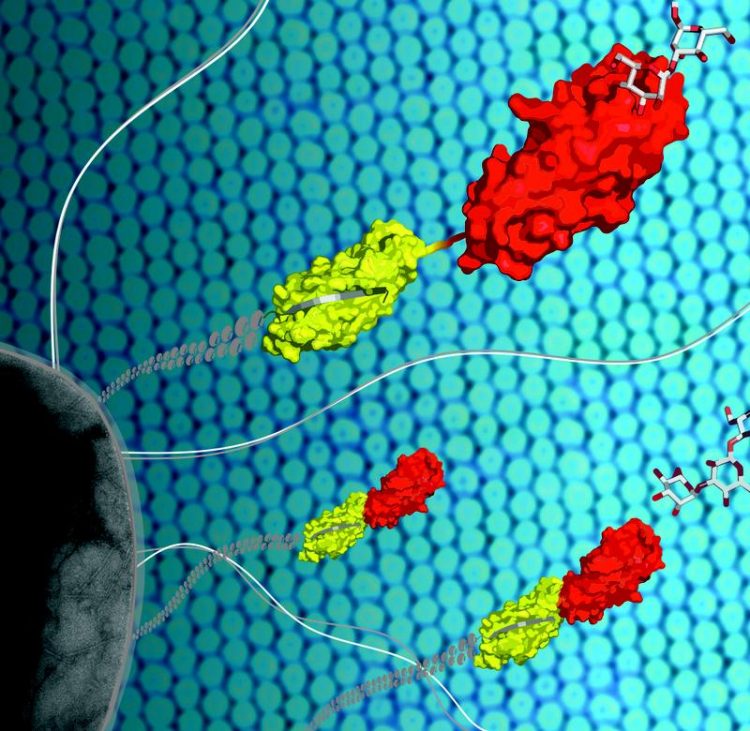
Using the protein FimH (yellow/red) located at the tip of long protrusions, the bacterial pathogen E. coli (grey) attaches to cell surfaces of the urinary tract. Maximilian Sauer, ETH Zürich
Many women have already experienced how painful a bladder infection can be: a burning pain during urination and a constant urge to urinate are the typical symptoms. The main cause of recurrent urinary tract infections is a bacterium found in the normal flora of the intestine, Escherichia coli. The bacteria enter the urinary tract, attach to the surface and cause inflammation.
The teams of Prof. Timm Maier at the Biozentrum and Prof. Beat Ernst at the Pharmazentrum of the University of Basel, along with Prof. Rudolf Glockshuber from the Institute of Molecular Biology and Biophysics at the ETH Zurich, have now discovered how bacteria adhere to the urinary tract under urine flow via the protein FimH and subsequently travel up the urethra.
Intestinal bacterium adheres to the cell surfaces with the protein FimH
The pathogen has long, hairlike appendages with the protein FimH at its tip, forming a tiny hook. This protein, which adheres to sugar structures on the cell surface, has a special property: It binds more tightly to the cell surface of the urinary tract the more it is pulled. As strong tensile forces develop during urination, FimH can protect the bacterium from being flushed out.
“Through the combination of several biophysical and biochemical methods, we have been able to elucidate the binding behavior of FimH in more detail than ever before”, says Glockshuber. In their study, the scientists have demonstrated how mechanical forces control the binding strength of FimH.
“The protein FimH is composed of two parts, of which the second non-sugar binding part regulates how tightly the first part binds to the sugar molecule“, explains Maier. “When the force of the urine stream pulls apart the two protein domains, the sugar binding site snaps shut. However, when the tensile force subsides, the binding pocket reopens. Now the bacteria can detach and swim upstream the urethra.”
Drugs against FimH to combat urinary tract infections
Urinary tract infections are the second most common reason for prescribing antibiotics. Yet, in times of increasing antibiotic resistance, the focus moves increasingly to finding alternative forms of treatment. For the prevention and therapy of E. coli infections, drugs that could prevent the initial FimH attachment of the bacteria to the urinary tract could prove to be a suitable alternative, as this would make the use of antibiotics often unnecessary.
This opens up the possibility of reducing the use of antibiotics and thus preventing the further development of resistance. Prof. Ernst, from the Pharmazentrum of the University of Basel, has been working intensively on the development of FimH antagonists for many years. The elucidation of the FimH mechanism supports these efforts and will greatly contribute to the identification of a suitable drug.
Original article
Maximilian M. Sauer, Roman P. Jakob, Jonathan Eras, Sefer Baday, Deniz Eriş, Giulio Navarra, Simon Bernèche, Beat Ernst, Timm Maier, Rudi Glockshuber
Catch-bond mechanism of the bacterial adhesin FimH
Nature Communications (2016), doi: 10.1038/ncomms10738
Further information
Prof. Dr. Timm Maier, University of Basel, Biozentrum, tel. +41 61 267 21 76, email:: timm.maier@unibas.ch
Prof. Dr. Rudolf Glockshuber, ETH Zürich, Institute of Molecular Biology & Biophysics, tel. +41 44 633 68 19, email: rudi@mol.biol.ethz.ch
Dr. Katrin Bühler, University of Basel, Biozentrum, Communications, tel. +41 61 267 09 74, email: katrin.buehler@unibas.ch
https://www.unibas.ch/en/News-Events/News/Uni-Research/Urinary-tract-infection-H…
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Superradiant atoms could push the boundaries of how precisely time can be measured
Superradiant atoms can help us measure time more precisely than ever. In a new study, researchers from the University of Copenhagen present a new method for measuring the time interval,…

Ion thermoelectric conversion devices for near room temperature
The electrode sheet of the thermoelectric device consists of ionic hydrogel, which is sandwiched between the electrodes to form, and the Prussian blue on the electrode undergoes a redox reaction…

Zap Energy achieves 37-million-degree temperatures in a compact device
New publication reports record electron temperatures for a small-scale, sheared-flow-stabilized Z-pinch fusion device. In the nine decades since humans first produced fusion reactions, only a few fusion technologies have demonstrated…





















