Special Antibodies Could Lead to HIV Vaccine
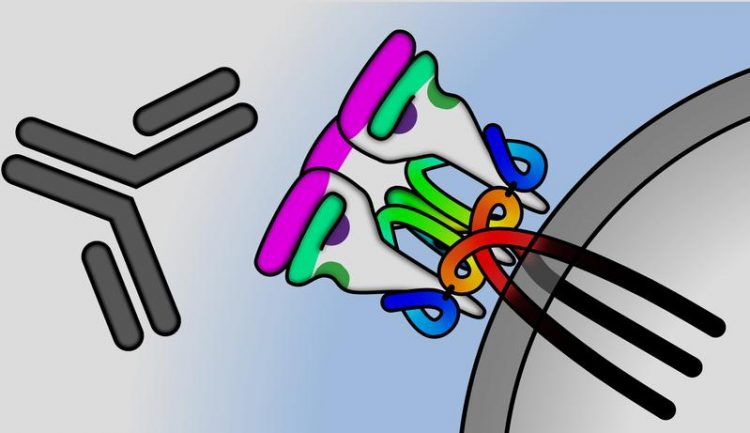
An antibody (dark grey) is binding to an HIV envelope protein (coloured) on the surface of the virus (light grey). UZH
A small number of people who are infected with HIV-1 produce very special antibodies. These antibodies do not just fight one virus strain, but neutralize almost all known virus strains. Research into developing an HIV vaccine focuses on discovering the factors responsible for the production of such antibodies.
HIV-1 genome influences immune reaction
A Swiss research team led by the University of Zurich (UZH) and University Hospital Zurich (USZ) has been searching for these factors for years. Several have already been identified: For example, the virus load and the diversity of the viruses, the duration of the infection, and the ethnicity of the affected person can all influence the body’s immune response. “In our new study, we were able to identify another factor: The genome of the HI virus,” says Huldrych Günthard, deputy director of the Department of Infectious Diseases and Hospital Epidemiology at USZ.
Antibody response of transmission pairs
The starting point for the researchers was the data and biobanked blood samples of around 4,500 HIV-infected people, recorded in the Swiss HIV Cohort Study and the Zurich Primary HIV Infection Study. In total the researchers found 303 potential transmission pairs – i.e. pairs of patients for whom the similarity of the viruses’ genomic RNA indicated that they were probably infected with the same virus strain. “By comparing the immune response of these pairs of patients, we were able to show that the HI virus itself has an influence on the extent and specificity of the antibody reactions,” explains the study’s first author Roger Kouyos, research group leader at the Department of Infectious Diseases and Hospital Epidemiology at USZ.
Special envelope proteins provide broad protection
Antibodies acting against HIV bind to proteins found on the surface of the virus. These envelope proteins differ according to virus strain and subtype. The researchers therefore examined more closely a patient pair with very similar virus genomes and at the same time very strong activity of broadly neutralizing antibodies. “We discovered that there must be a special envelope protein that causes an efficient defense,” explains Alexandra Trkola, virologist and head of the Institute of Medical Virology at UZH.
Search for an ideal envelope protein continues
In order to be able to develop an effective vaccine against HIV-1, it is necessary to pinpoint the envelope proteins and virus strains that lead to the formation of broadly acting antibodies. It is therefore planned to widen the search. “We have found one candidate. Based on that, we now want to begin developing an immunogen ourselves,” adds Trkola.
Prof. Alexandra Trkola, PhD
Institute of Medical Virology
University of Zurich
Phone: +41 44 634 53 80
E-mail: trkola.alexandra@virology.uzh.ch
Prof. Roger Kouyos, PhD
Department of Infectious Diseases and Hospital Epidemiology
UniversityHospital Zurich
Phone: +41 44 255 36 10
E-mail: roger.kouyos@usz.ch
Prof. Huldrych Günthard, M.D.
Department of Infectious Diseases and Hospital Epidemiology
UniversityHospital Zurich
Phone: +41 44 255 34 50
E-mail: huldrych.guenthard@usz.ch
Roger D. Kouyos, et. al. Tracing HIV-1 strains that imprint broadly neutralising antibody responses. Nature. September 10, 2018. DOI: 10.1038/s41586-018-0517-0
http://www.media.uzh.ch/en/Press-Releases/2018/HIV-Vaccine.html
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Zap Energy achieves 37-million-degree temperatures in a compact device
New publication reports record electron temperatures for a small-scale, sheared-flow-stabilized Z-pinch fusion device. In the nine decades since humans first produced fusion reactions, only a few fusion technologies have demonstrated…
Innovative microscopy demystifies metabolism of Alzheimer’s
Researchers at UC San Diego have deployed state-of-the art imaging techniques to discover the metabolism driving Alzheimer’s disease; results suggest new treatment strategies. Alzheimer’s disease causes significant problems with memory,…
A cause of immunodeficiency identified
After stroke and heart attack: Every year, between 250,000 and 300,000 people in Germany suffer from a stroke or heart attack. These patients suffer immune disturbances and are very frequently…





















