Shedding light on an assistant protein
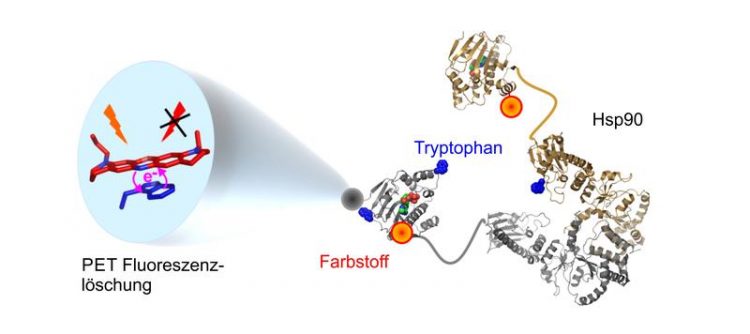
The combination of a dye molecule and tryptophan opens up wholly new insights into the movements of the protein Hsp90. Graphic: Hannes Neuweiler
Proteins are among the functional key elements of life. Made up of long chains of amino acids, they fold to form highly organised, three-dimensional structures much like an origami creation. However, folded proteins are not stiff, but variable and in constant motion. This allows them to act as molecular machines that execute a variety of functions which together create life.
Errors due to packing in a tight space
Living cells are full of such proteins which, however, frequently get in each other's way when they fold or perform their functions. “Errors in folding or functioning within these tightly packed quarters can trigger a number of diseases and even cancer,” explains Dr. Hannes Neuweiler, group manager at the Department of Biotechnology & Biophysics of the University of Würzburg. Together with his work group he has developed a technique that enables proteins at work to be observed with high spatial and temporal resolution. The group presents the results of its work in the latest issue of the journal Nature Chemical Biology.
Neuweiler and his team have focused on so-called chaperones. These are proteins that help other proteins to fold, activate them through reshaping and prevent undesired clustering,” Neuweiler explains.
A heat shock protein with healing powers
The heat shock protein Hsp90 is an exceptional specimen of such specialised proteins: It is one of the most frequently occurring proteins in living cells where it assists a huge number of “patients” of various shapes and purposes. “The healing power of Hsp90 is, however, mysterious. Up to now, its exact function has been known only in part,” says Neuweiler.
What was known is that the chaperone is similar to a molecular bracket that opens and closes while attending to its patients. In the past, researchers used crystallographic methods and X-ray diffraction to determine atomically resolved structures of Hsp90 that show snapshots from the chaperone's engine room. “It had, however, been impossible to observe this mechanism of Hsp90 when working in aqueous solution,” Neuweiler explains. There were no methods available to visualize such local motions in proteins.
A beacon highlights structural changes
This has changed recently: The Würzburg scientists have developed high-resolution fluorescence probes that allow observing these motions in Hsp90. Like a beacon being switched on and off when structures change, the probes indicate when and on which time scale a motion takes place in the molecular machine.
The researchers have harnessed the phenomenon of quenching through photo-induced electron transfer (PET) for this purpose. The principle: Synthetic dye molecules that emit light under normal circumstances are switched off by a photochemical reaction when in contact with the naturally occurring amino acid tryptophan. Neuweiler and his co-workers have equipped the chaperone with “motion sensors” by inserting such dye molecules at selected spots in Hsp90 in the vicinity of tryptophan. The results of their work show that local structural elements in Hsp90 move synchronously while the molecular bracket opens and closes. The cooperating protein Aha1, a so-called co-chaperone, operates the lever of a selected structural element of Hsp90 in an early phase, thereby accelerating the process.
In future, the scientists plan to shed light on other structural changes in Hsp90 and the functioning of other co-chaperones using the new fluorescence technology. They hope to glean new insight into the workings of assistant proteins and thus on the evolution of diseases from their investigations of single molecules using sensitive imaging techniques.
“Cooperation of local motions in the Hsp90 molecular chaperone ATPase mechanism”, Andrea Schulze, Gerti Beliu, Dominic A. Helmerich, Jonathan Schubert, Laurence H. Pearl, Chrisostomos Prodromou & Hannes Neuweiler. Nature Chemical Biology
Contact
Dr. Hannes Neuweiler, phone: +49 931 31-83872, e-mail: hannes.neuweiler@uni-wuerzburg.de
http://dx.doi.org/10.1038/nchembio.2111 The Paper in Nature Chemical Biology
Media Contact
All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

High-energy-density aqueous battery based on halogen multi-electron transfer
Traditional non-aqueous lithium-ion batteries have a high energy density, but their safety is compromised due to the flammable organic electrolytes they utilize. Aqueous batteries use water as the solvent for…

First-ever combined heart pump and pig kidney transplant
…gives new hope to patient with terminal illness. Surgeons at NYU Langone Health performed the first-ever combined mechanical heart pump and gene-edited pig kidney transplant surgery in a 54-year-old woman…

Biophysics: Testing how well biomarkers work
LMU researchers have developed a method to determine how reliably target proteins can be labeled using super-resolution fluorescence microscopy. Modern microscopy techniques make it possible to examine the inner workings…





















