Phenotype at the push of a button
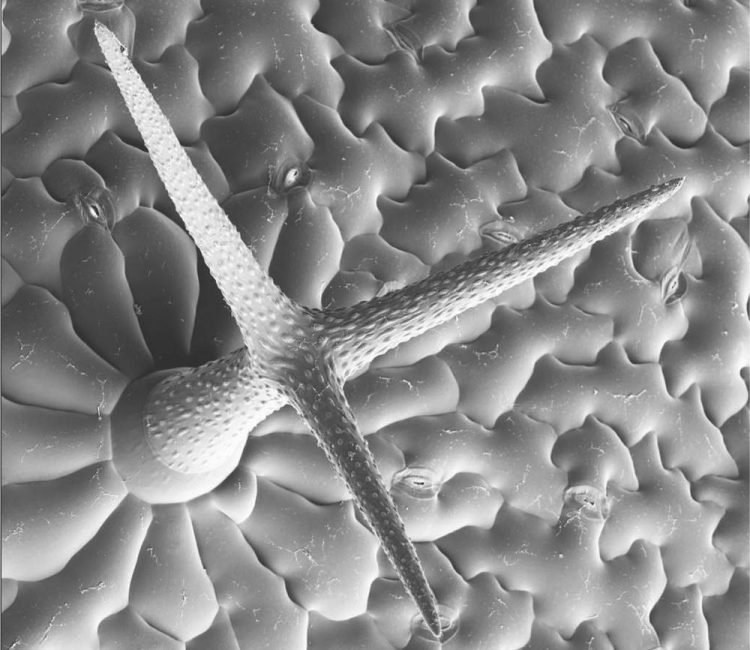
Trichome on a leaf of Arabidopsis. Foto: Nico Dissmeyer, IPB
Scientists at the Leibniz Institute of Plant Biochemistry (IPB) in Halle have developed a method that makes it possible to accumulate or break down chosen proteins in living organisms as needed. In order to accomplish this, researchers around Dr. Nico Dissmeyer together with scientists from Cologne and Zurich have developed a molecular switch that can be activated by temperature changes.
This switch enables plants to produce the desired protein in large quantities at low ambient temperatures. On the other hand, the protein is completely broken down within a few hours after a moderate temperature increase. As a result, the exterior appearance of plants – the phenotype – can be changed for the first time through a very tightly regulated intervention for a limited time directly on protein levels.
However, the process is not only applicable to various plants but has also been successfully used in animal cell cultures, baker’s yeast and even living fruit flies. Various possible applications in fundamental research and biotechnology production methods are conceivable. Now the method has been published in Nature Communications magazine.
The temperature shift method is based on the natural protein disposal machinery (the proteasome) that occurs in every living cell, from baker’s yeast to humans. All proteins consistently have to act in the right place, at the right time and with the correct activity, especially those that – as enzymes – regulate metabolic reactions and therefore important physiological processes such as breathing, digestion, development and immune defence. Defective enzymes that are over- or underactive, or are active for too long or not at all, can lead to serious illnesses for the organism. Therefore, they are identified as abnormal in the cells and broken down.
The Halle scientists used this natural process to accumulate or break down proteins of their choice in the cell depending on the ambient temperature. Artificial DNA constructs consisting of two gene sequences were introduced into the plant cell for this purpose. The first gene encoded for a temperature-sensitive protein that formed a defective conformation with an increase in the ambient temperature. The second gene contained the information for the respective desired protein to be accumulated in the cells on demand. These genes formed the basis for the biosynthesis of a fusion protein within the cell that accumulated in the cell at low ambient temperatures of 13°C and carried out its function correctly.
After a temperature shift to 29°C, the temperature-sensitive element of the fusion protein changed its conformation so that it was identified as abnormal by the proteasome and therefore broken down. The desired protein as a fixed component of the fusion protein was degraded as well. In this system, the temperature-sensitive component of the fusion protein therefore serves as a molecular temperature switch. With a temperature increase, gradual or for a limited time, it was even possible to gradually decrease the quantity of the desired protein in this manner and the change was also reversible. After reducing the ambient temperature, the desired protein was again accumulated in the cells.
Nico Dissmeyer and his colleagues successfully tested the temperature shift method in different organisms with various target proteins. In the genetic model system mouse-ear cress (Arabidopsis thaliana), they were able to trigger the development of small, single-cell hairs on the leaf surface (trichomes) by using the technology. Here a gene required for trichome development in combination with the molecular switch gene was transferred to a mutant that generally does not form any of these trichome cells. As a result, the plant formed trichomes on its leaves again at an ambient temperature of 13°C while the trichome formation protein was deactivated at 29°C, so that all leaves were glabrous and hairless, exactly as in the defective null mutant.
Trichome formation, in particular when achieved in an ‘on demand’ manner, harbours great potential for applications of the temperature shift in biotechnology production processes. Certain plants form trichomes on their leaves, serving as glandular hairs. These glandular trichomes produce and store plant metabolism products such as essential oils or antibodies against harmful insects. They form a self-contained cell system that is not connected to the plant’s vascular system.
That is why heavy metals and other catabolic waste products that are toxic to the plant are often accumulated and stored in the glandular hairs. With the temperature shift method, trichomes can be used as microreactors for the specific, controlled production of proteins toxic to the plant or for other active substances. It would for example be possible to use trichomes as miniature factories for pharmaceutical and other high-value products. Tobacco plants with their large leaves and correspondingly large numbers of glandular hairs would be especially well suited for this method.
Nico Dissmeyer is head of the independent junior research group ‘protein recognition and degradation’ at the Leibniz Institute of Plant Biochemistry since 2011. Since the end of 2011, he is being financed as junior research group leader of the ‘ScienceCampus Halle – Plant-Based Bioeconomy’ by the state of Saxony-Anhalt, the Leibniz Association, the EU and the Deutsche Forschungsgemeinschaft. With the temperature shift method, his workgroup makes a powerful molecular tool available to the life sciences in order to change certain characteristics of organisms quickly, subtly nuanced and reversibly.
Original publication:
Frederik Faden, Thomas Ramezani, Stefan Mielke, Isabel Almudi, Knud Nairz, Marceli S. Froehlich, Jörg Höckendorff, Wolfgang Brandt, Wolfgang Hoehenwarter, R. Jürgen Dohmen, Arp Schnittger & Nico Dissmeyer, Phenotypes on demand via switchable target protein degradation in multicellular organisms. Nature Communications 7: 12202, doi:10.1038/ncomms12202
http://www.nature.com/ncomms/2016/160721/ncomms12202/full/ncomms12202.html
http://www.ipb-halle.de/en/public-relations/news/article-detail/phaenotyp-auf-kn…
Media Contact
More Information:
http://www.ipb-halle.de/All latest news from the category: Life Sciences and Chemistry
Articles and reports from the Life Sciences and chemistry area deal with applied and basic research into modern biology, chemistry and human medicine.
Valuable information can be found on a range of life sciences fields including bacteriology, biochemistry, bionics, bioinformatics, biophysics, biotechnology, genetics, geobotany, human biology, marine biology, microbiology, molecular biology, cellular biology, zoology, bioinorganic chemistry, microchemistry and environmental chemistry.
Newest articles

Superradiant atoms could push the boundaries of how precisely time can be measured
Superradiant atoms can help us measure time more precisely than ever. In a new study, researchers from the University of Copenhagen present a new method for measuring the time interval,…

Ion thermoelectric conversion devices for near room temperature
The electrode sheet of the thermoelectric device consists of ionic hydrogel, which is sandwiched between the electrodes to form, and the Prussian blue on the electrode undergoes a redox reaction…

Zap Energy achieves 37-million-degree temperatures in a compact device
New publication reports record electron temperatures for a small-scale, sheared-flow-stabilized Z-pinch fusion device. In the nine decades since humans first produced fusion reactions, only a few fusion technologies have demonstrated…





















